Discovering potential therapeutic protein inhibitors for Chagas disease
Chagas disease is a potentially life-threatening illness caused by the parasite Trypanosoma cruzi, and is transmitted to humans through triatomine blood-sucking insects commonly referred to as "kissing bugs" or "vampire bugs." While it was once confined to the Americas, worldwide travel has spread the disease, which is now endemic to approximately 20 countries. Currently, the World Health Organization (WHO) estimates that 10 to 13 million people are chronically infected, around 90 million people are exposed to the risk of the infection, and nearly 21,000 people die each year as a result, making effective treatment a necessity. Current treatments are largely effective in the acute phase of the infection but have significantly diminished efficacy in the chronic phase of Chagas disease. Moreover, these drugs, which were developed in the 1960s, are associated with severe adverse effects.
Masakazu Sekijima of Nagasaki University and colleagues have used a multi-modal integrated approach to develop potential new anti-Chagas therapies by combining the principles of structure-based drug design using knowledge of the target's three-dimensional (3-D) structure, with in vitro testing methods and X-ray crystallography. This approach narrows the range of potential drug candidates more efficiently. Through virtual screening by the TSUBAME supercomputer at Tokyo Tech, they selected their target protein, T. cruzi spermidine synthase, based on specific structural features and properties indicating its importance for survival in another Trypanosoma species. If the protein is required for survival of a species, inhibiting that protein could be a potential mechanism of action for a drug with activity against the parasite that causes Chagas disease.
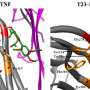
They focused on Spermidine synthase (SpdSyn) as the target protein, as sourced from the iNTRODB system. The in-house web system iNTRODB facilitates the selection of drug target proteins for NTDs, particularly for trypanosomiasis. This system provides information on trypanosomal proteins with useful annotations, including the protein structure from the Protein Data Bank (PDB) and the protein inhibitors from ChEMBL.
Following selection, potential drug candidate inhibitors were identified through a screening search known as docking simulation—a structure-based drug design approach using 3-D simulations to computationally match drug compounds to SpdSyn. They successfully identified four drug-like compounds that were virtual "matches" (referred to as "hits"), then evaluated their inhibition activity in vitro and compared the results with those of a positive control. To further test potential activity and binding, they employed X-ray crystallography to confirm these four compounds in complex with the protein structure. Through interaction analyses for each compound, the researchers found that all four compounds interacted with the proposed target binding sites through the same amino acid, Asp171. Additionally, molecular simulation suggested additional interacting sites for each compound that was not predicted by docking simulation.
Sekijima and team believe that their study's findings are indicative of the promise that docking simulation holds for the identification of potential drug-like inhibitors of the target protein and therapies for Chagas disease. They hope to demonstrate the general applicability of their approach, opening doors to the discovery of treatments for other diseases.
More information: Ryunosuke Yoshino et al, In silico, in vitro, X-ray crystallography, and integrated strategies for discovering spermidine synthase inhibitors for Chagas disease, Scientific Reports (2017). DOI: 10.1038/s41598-017-06411-9