October 19, 2017 report
Cancer drug found to offer promising results in treating sepsis in test mice
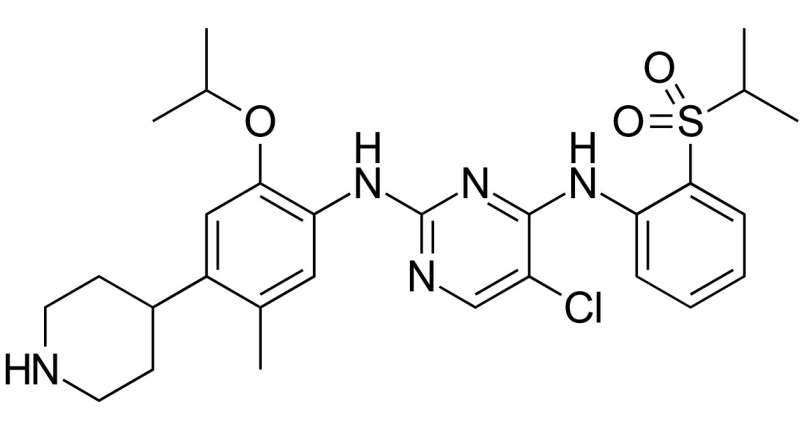
(Medical Xpress)—A combined team of researchers from China and the U.S. has found that a drug commonly used to treat lung cancer in humans offers a degree of protection against sepsis in test mice. In their paper published in Science Translational Medicine, the team describes their study of the cause of sepsis, their search for a therapy to decrease its occurrence, and the results of testing ceritinib as as a therapy in mice.
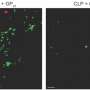
Sepsis is a condition in which bacteria or other infectious organisms flood the bloodstream, causing a host of problems and, in many cases, death. It is also known as blood poisoning or septicemia, and is a leading cause of death in most modern hospitals. For that reason, health scientists have dedicated a lot of time and effort into learning about the factors that cause it, how to prevent it, and treating it when it does. Prior research has shown that one of the main reasons it is so deadly is because the immune system goes into overdrive when confronting the infection with calamitous results including cascading organ failure. In this new effort, the researchers took a closer look at a stimulator of interferon genes (STING), which activates prior to the onset of sepsis and which is believed to be among the agents that cause sepsis to kick off. They wondered if preventing STING from activating might prevent sepsis from occurring. To find out, they embarked on a search for drugs that might do the trick. They found one in particular that seemed promising: ceritinib, known also as the commercial drug Zykadia, used to treat some types of lung cancers.
Ceritinib, the researchers note, does its work by suppressing a protein called ALK, which serves as a signal to set off a STING response. The researchers tested the drug by giving it to test mice that were on the path to developing sepsis and report doing so showed promising results. It protected some of the test animals against sepsis, which in turn prevented organ damage. A lot more testing will need to be done to find out if ceritinib can be used to protect humans from sepsis, including testing it in animals that are farther along the path to sepsis, and whether its effectiveness is tied to particular infections.
More information: Ling Zeng et al. ALK is a therapeutic target for lethal sepsis, Science Translational Medicine (2017). DOI: 10.1126/scitranslmed.aan5689
Abstract
Sepsis, a life-threatening organ dysfunction caused by infection, is a major public health concern with limited therapeutic options. We provide evidence to support a role for anaplastic lymphoma kinase (ALK), a tumor-associated receptor tyrosine kinase, in the regulation of innate immunity during lethal sepsis. The genetic disruption of ALK expression diminishes the stimulator of interferon genes (STING)–mediated host immune response to cyclic dinucleotides in monocytes and macrophages. Mechanistically, ALK directly interacts with epidermal growth factor receptor (EGFR) to trigger serine-threonine protein kinase AKT phosphorylation and activate interferon regulatory factor 3 (IRF3) and nuclear factor κB (NF-κB) signaling pathways, enabling STING-dependent rigorous inflammatory responses. Moreover, pharmacological or genetic inhibition of the ALK-STING pathway confers protection against lethal endotoxemia and sepsis in mice. The ALK pathway is up-regulated in patients with sepsis. These findings uncover a key role for ALK in modulating the inflammatory signaling pathway and shed light on the development of ALK-targeting therapeutics for lethal systemic inflammatory disorders.
© 2017 Medical Xpress