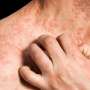
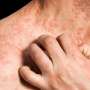
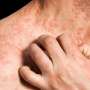
Crisaborole safe for long-term treatment of atopic dermatitis
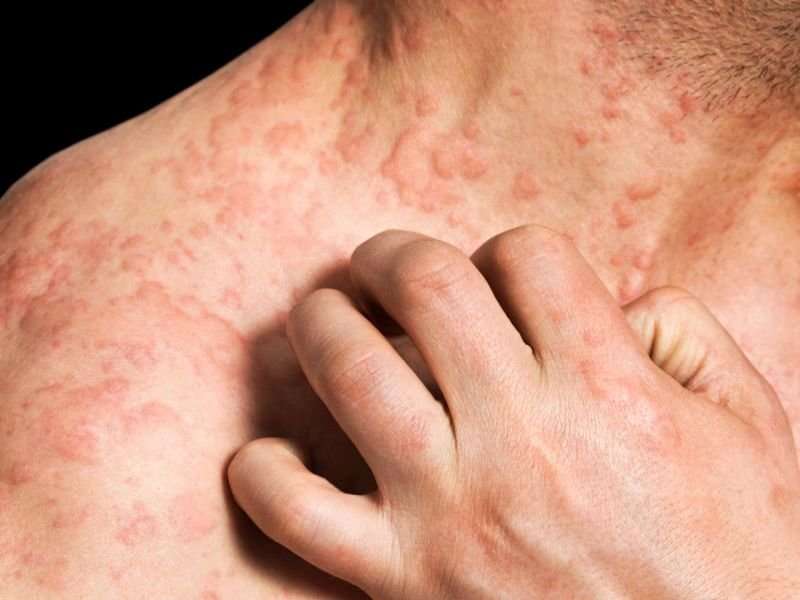
(HealthDay)—Crisaborole ointment appears to be safe for the long-term treatment of atopic dermatitis (AD), according to a study published in the October issue of the Journal of the American Academy of Dermatology.
Lawrence F. Eichenfield, M.D., from Rady Children's Hospital in San Diego, and colleagues assessed the long-term safety results from a multicenter, open-label, 48-week safety study of 517 patients ≥2 years of age with mild to moderate AD who continued crisaborole treatment after completing a 28-day phase 3 pivotal study.
The researchers found that during the pivotal study and the open-label trial, 65 percent of patients reported at least one treatment-emergent adverse event (TEAE), most of which were mild (51.2 percent) or moderate (44.6 percent) and considered unrelated to treatment (93.1 percent). There was consistency with regard to the frequency and severity of TEAEs. Overall, treatment-related AEs were reported in 10.2 percent of patients and most commonly included dermatitis atopic (3.1 percent), application-site pain (2.3 percent), and application-site infection (1.2 percent). Because of TEAEs, nine patients (1.7 percent) discontinued the long-term study.
"Crisaborole ointment had a low frequency of treatment-related AEs over 48 weeks of treatment of patients with AD," conclude the authors.
Several authors disclosed financial ties to the pharmaceutical industry, including Anacor Pharmaceuticals, which funded the study.
More information: Abstract/Full Text
Copyright © 2017 HealthDay. All rights reserved.