Researchers discover an Achilles heel in a lethal leukemia

Researchers have discovered how a linkage between two proteins in acute myeloid leukemia enables cancer cells to resist chemotherapy and showed that disrupting the linkage could render the cells vulnerable to treatment. St. Jude Children's Research Hospital scientists led the research, which could lead to drugs to enhance chemotherapy in patients with AML, colon and breast cancers and the brain tumor medulloblastoma.
New treatments for AML are critical because overall five-year survival for adults and children with this leukemia is about 27 percent. John Schuetz, Ph.D., a member of the St. Jude Department of Pharmaceutical Sciences, led the research, which appears today in the scientific journal Nature Communications.
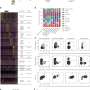
The investigators launched their experiments based on findings from other scientists that a protein called ABCC4 was greatly elevated in aggressive cases of AML. Schuetz and his colleagues searched for other proteins that might interact with ABCC4 and enable its function. Their screening of hundreds of candidate proteins yielded one, MPP1, which was also greatly increased in AML.
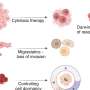
The researchers showed that the two proteins were connected and that the connection enabled cells to assume the characteristics of highly proliferative leukemia cells. These experiments involved genetically altering blood-forming blood progenitor cells to have high MPP1 and ABCC4 levels. The cells were grown in culture and then transferred, or replated, to new culture dishes to see if the cells would continue to grow. Such self-renewal is a hallmark of leukemia cells. The researchers found that serial regrowth depended on the cells having high levels of both ABCC4 and MPP1.
"Typically if you take normal progenitors and you replate, you could do that one time, maybe twice," Schuetz said. "But our big surprise was that overexpressing MPP1—analogous to what you would see in leukemia—allows those progenitors to self-renew, to be replated over and over, to form new colonies."
The experiments also revealed that the two proteins functioned at the cell membrane, where they could play a role in the machinery that would rid the cancer cells of chemotherapy drugs.
"When we disrupted their interaction, ABCC4 moved off the membrane and the cells became more sensitive to drugs used in AML—drugs that are pumped out of the cell by ABCC4," Schuetz said.
By screening thousands of compounds, the researchers identified those that could disrupt the ABCC4-MPP1 connection. One, called Antimycin-A, reversed drug resistance in AML cell lines and in cells from AML patients. Antimycin-A is too toxic to be used in chemotherapy, but identification of the compound should help scientists searching for other, less-toxic drugs to disrupt the ABCC4-MPP1 interaction.
The findings could also enable clinicians to identify AML patients with high levels of ABCC4 and MPP1. Such patients might benefit from drugs that disrupt ABCC4-MPP1 by enhancing the effectiveness of standard chemotherapy, Schuetz said. Also, he noted, other cancers including breast and colon cancer and medulloblastoma show high levels of both ABCC4 and MPP1. Chemotherapy for those cancers might also be enhanced by drugs that disrupt ABCC4-MPP1.
More information: Aaron Pitre et al. An unexpected protein interaction promotes drug resistance in leukemia, Nature Communications (2017). DOI: 10.1038/s41467-017-01678-y