Research in mice paves way to teasing out cause and effect between gut microbes and disease
Clearing a major hurdle in the field of microbiome research, Harvard Medical School scientists have designed and successfully used a method to tease out cause-and-effect relationships between gut bacteria and disease.
Reporting Dec. 6 in Nature, the team says the approach could propel research beyond mere microbiome-disease associations and elucidate true cause-effect relationships.
The experiments, conducted in mice, also identify a previously unknown gut microbe that tames intestinal inflammation and protects against severe colitis. The researchers say the finding makes a strong case for testing the newly identified gut bacterium as a probiotic therapy in people with inflammatory bowel disease, a constellation of conditions marked by chronic inflammation of the intestines and estimated to affect up to 1.3 million people in the United States, according to the Centers for Disease Control and Prevention.
The approach uses a sort of "microbial triangulation." It mimics the principles of classic maritime navigation or, in more modern terms, tracking the location of a mobile phone by verifying data from multiple sources—but instead of stars or cell phone towers, the researchers are homing in on intestinal bugs. Based on the method of elimination, the technique involves the gradual narrowing down of bacterial species to identify specific microbes that modulate the risk for specific diseases. In the current study, researchers adapted the principles to identify beneficial, protective bacteria.
"Our approach can help scientists find the proverbial needles in a 'haystack' of thousands of microbes that are currently thought to modulate health," said investigator Dennis Kasper, professor of microbiology and immunobiology at Harvard Medical School. "If the field is to move past associations—the Achilles' heel in microbiome research—we need a system that reliably teases out causative relationships between gut bacteria and disease. We believe our method achieves that," added Kasper, who is also the Harvard Medical School William Ellery Channing Professor of Medicine at Brigham and Women's Hospital.
Over the last decade, study after study has identified thousands of commensal microbes—those residing innocently in our bodies—and catalogued observations of possible links between groups of microbes and the presence or absence of a panoply of diseases, including diabetes, multiple sclerosis and inflammatory bowel disease. Yet, scientists don't know whether and how the presence of specific microbes—or fluctuations in their numbers—affects health. It remains unclear whether certain microbes are innocent bystanders, mere markers of disease, or whether they are active agents, causing harm or providing protection against certain ailments.
The holy grail of this work would be not to merely define whether a microbe fuels or minimizes the risk for a given disease but to discover microbes and microbial molecules that can be used therapeutically.
"The ultimate goal is to clarify the mechanisms of disease and then identify bacterial molecules that can be used to treat, reverse or prevent it," said study lead author Neeraj Surana, Harvard Medical School instructor in pediatrics and an infectious disease specialist at Boston Children's Hospital.
Old-fashioned detective work
For their study, Kasper and Surana compared the gut microbiomes of several groups of mice that harbored different populations of intestinal bacteria.
The researchers started out with two groups of mice. One group had been bred with human gut microbiomes—housing intestinal bacteria normally found in human intestines. The other group had been bred to harbor normal mouse microbiomes. When researchers gave the animals a chemical compound that triggered intestinal inflammation, or colitis, mice that harbored human intestinal microbes were protected from the effects of the disease. Mice whose guts harbored typical mouse bacteria, however, developed severe symptoms.
Next, the researchers housed all mice in the same living space. Sharing living space for as briefly as one day led to noticeable changes in how the animals responded to disease. Mice that had been originally protected from colitis started showing more serious signs of it, while colitis-prone mice grew increasingly resistant to the effects of the condition and developed milder symptoms—a proof-of-principle finding which shows that exchange of intestinal bacteria through shared living space can lead to changes in the animals' ability to cope with the disease.
The needle in the haystack
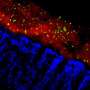
The disease-modulating microbe would be lurking amid the hundreds of bacterial species present in all mice. But given that each mouse group harbored between 700 and 1,100 bacterial species in their guts, how could scientists identify the one that truly mattered in colitis? The team began by analyzing the intestinal makeup of each one of the mouse groups, comparing their microbial profiles before and after they shared a living space. To "triangulate" the suspect's identity, scientists looked for microbes that were either scarce or abundant, tracking with colitis severity. In other words, the numbers of the causative microbe would either go up or down with disease severity, the scientists reasoned. Only one such microbial group fit the profile—a bacterial family known as Lachnospiraceae, commonly found in human intestines as well as the guts of other mammals.
To pinpoint the one organism within the Lachnospiraceae family that regulates response to colitis, the researchers isolated one bacterial species and gave it to colitis-prone mice. To compare its effects against other microbes, they also gave the animals organisms from different bacterial families. The only bacterium that protected colitis-prone animals from the ravages of the disease was a never-before-described microbe that the researchers had isolated from the guts of mice seeded with human feces, the animals that had harbored human microbiomes. The microbe was notably absent from mice with mouse microbiomes. Because of its immune-protective properties, Kasper and Surana christened the newly identified organism Clostridium immunis.
The isolation of the disease-modifying microbe makes a powerful case for testing it as therapy in people with inflammatory bowel disease, the researchers said.
Taken together, the team said, the experiments show that a model of winnowing the list of possible microbial suspects down to the level of individual species is not only feasible but critical in unmasking specific disease-modulating microbes.
More information: Moving beyond microbiome-wide associations to causal microbe identification, Nature (2017). nature.com/articles/doi:10.1038/nature25019