Scientists analyze viability of shRNA therapy for Huntington's Disease
Researchers from the Biogerontology Research Foundation , Department of Molecular Neuroscience at the Swammerdam Institute for Life Sciences at the University of Amsterdam, and the Department of Neurobiology, Care Sciences and Society at the Karolinska Institute (ki.se/en/startpage) announce the publication of a paper in Translational Neurodegeneration, a BioMedCentral journal, titled RNAi mechanisms in Huntington's disease therapy: siRNA versus shRNA.
After many years of development, RNAi therapeutics are nearing the clinic. There are several variants on RNAi therapeutics, such as antisense oligonucleotides (ASOs), short-hairpin RNA (shRNA), small interfering RNA (siRNA), et cetera. The researchers' paper aimed to answer the question of why RNAi therapeutics for nucleotide repeat disorders (specifically Huntington's, a devastating genetic neurodegenerative disease), have lost favor in recent years. After a phenomenal amount of excitement, these therapies were hindered by problems like molecular stability, dosing, and transcriptional control of the gene therapeutic construct.
"We compared various RNAi-based therapeutic modalities available for the treatment of Huntington's Disease and offered mechanistic proposals on how to break through current barriers to clinical development. One key problem has proven to be modulating the expression level of shRNA constructs, which would otherwise be the clear frontrunner among ASOs, siRNAs, and other methods due to duration of expression, dramatically reduced off-target effects, and ease of delivery by adeno-associated viruses that are already approved by the EMA and FDA. We also put forward novel methods of modulating construct expression and avoiding off-target effects" said Franco Cortese, co-author of the paper and Deputy Director of the Biogerontology Research Foundation.
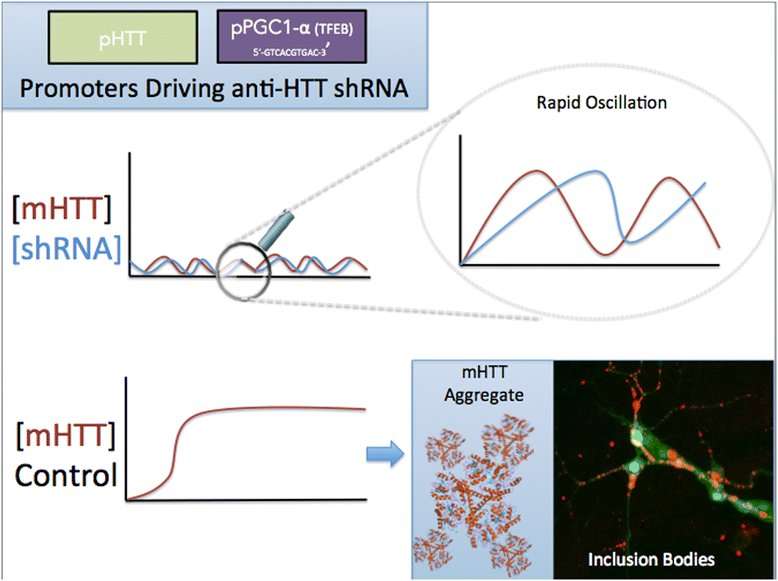
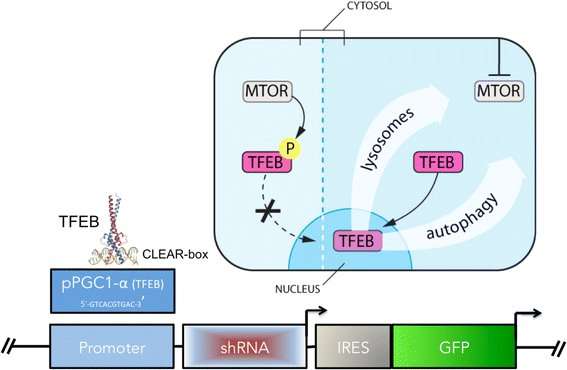
The researchers analyzed available data on the levels of off-target effects associated with siRNA vs shRNA, surveyed emerging strategies to reduce off-target effects in shRNA therapies (such as tough decoy RNAs, or TuDs), and proposed novel methods of controlling shRNA expression, in particular through the use of negative feedback-driven oscillating promoters.
"We proposed two novel feedback mechanisms that 1) activate construct expression stoichiometrically with mutant Huntingtin expression, or 2) only during aggregate-induced autophagy and lysosomal biogenesis. That way, the problem of excessive construct expression may be mitigated. These ideas were inspired by feedback systems used in synthetic biology, and in 'nonsynthetic,' naturally occurring biological systems" said Sebastian Aguiar, lead author of the paper.
More information: Sebastian Aguiar et al, RNAi mechanisms in Huntington's disease therapy: siRNA versus shRNA, Translational Neurodegeneration (2017). DOI: 10.1186/s40035-017-0101-9















