Test detects prior infection by Zika virus
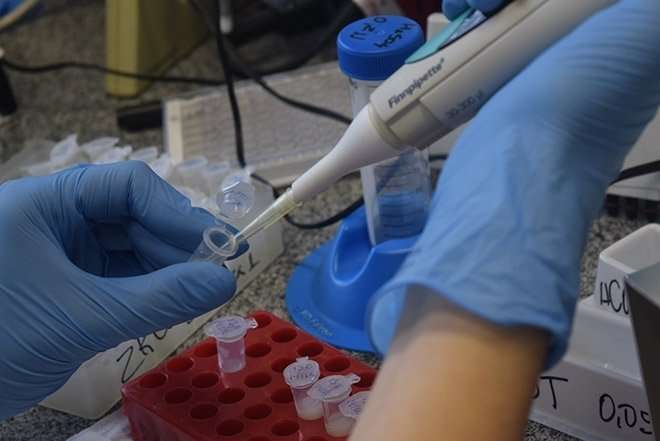
A test that detects antibodies against Zika virus in samples of blood serum with high specificity—and hence a low risk of cross-reaction with related microorganisms such as dengue virus—is set to enter the Brazilian market before 2018 is out.
The method was designed by Brazilian scientists. Technology firm Inovatech, with the support from the São Paulo Research Foundation—FAPESP through its Innovative Research in Small Business Program (PIPE), developed the test in collaboration with scientists at the University of São Paulo's Biomedical Science Institute (ICB-USP) and at the Butantan Institute.
"We're in the final stages of developing a serological test to detect immunoglobulin G (IgG) antibodies, which remain in the organism for many years after infection, providing lifelong immunity. We expect it to be approved for sale in the second half of this year," said Danielle Bruna Leal de Oliveira, a researcher at ICB-USP's Clinical & Molecular Virology Laboratory and principal investigator for the project.
Since the creation in 2016 of the Zika Virus Research Network in São Paulo (Rede Zika), which is supported by FAPESP, developing this type of diagnostic method has been considered a priority.
A tool of this kind, according to the scientists, is essential for answering several strategic questions for any plan of action against the disease: What is the exact size of the epidemic (distinguishing more precisely between cases of dengue and Zika, in both current and past outbreaks)? What proportion of pregnant women are infected (and hence immune)? How many women risk giving birth to babies with neurological problems due to congenital infection?
"The idea is to include this test in the prenatal examination process," Oliveira said. "If we can do that, pregnant women with a negative result [meaning they have never been infected] will take more precautions, such as avoiding travel to high-risk areas and using insect repellent. Meanwhile, those who are immune will be able to stop worrying."
According to Oliveira, currently available diagnostic tests either work only in the acute phase of infection—as is the case with the molecular method (real-time PCR) and chromatography, which detect viral RNA in the bloodstream—or detect Zika antibodies with low specificity.
"The specificity of the serological tests currently on the market falls within the 69%-75% range. So there's at least a 25% chance of a false positive if the patient was infected by dengue virus in the past. Our test's specificity is 93% for Zika," Oliveira said.
Like its predecessors, the test developed by Inovatech is based on a methodology known as ELISA, an acronym for enzyme-linked immunosorbent assay. The platform consists of a microtiter plate with 96 tiny wells to which adheres a viral protein recognized by the human immune system.
The wells are filled with blood serum from up to 94 patients, with two or more being reserved for controls. In the case of prior contact with Zika, IgG antibodies adhere to the viral protein, which is later detected by a colorimetric assay (positive and negative samples acquire different colorings).
"One of the difficulties associated with the method is that the viral protein commonly used, NS1, is very similar in all flaviviruses [the family Flaviridae includes dengue, Zika and yellow fever, among others]. To solve this problem, we used an edited version of the protein, selecting only the most Zika-specific part of the molecule," Oliveira explained. This "truncated" version of NS1 (δNS1) was developed by Luis Carlos de Souza Ferreira and his team at ICB-USP.
To reduce the risk of cross-reaction with dengue virus still further, Inovatech's researchers added another stage to the assay. Before the blood serum from patients is placed in the ELISA plate in contact with δNS1, it is exposed to proteins from dengue virus, with the aim of extracting all antibodies against this pathogen from the samples.
"The drawback is that it takes a little longer to get a result—about three hours, compared with two hours and twenty minutes for conventional ELISA—but we're working on a way to make it faster. The goal is that it should be quicker than the standard method because it's designed for use by hospitals in patient screening," Oliveira said.
Low cost
Selected in a call for proposals (in Portuguese) issued in 2016 under the aegis of an agreement between FAPESP and FINEP, the Brazilian Innovation Agency, the project coordinated by Oliveira aims to create a fast, low-cost serological test to detect both IgG and IgM (immunoglobulin M), which stays in the organism for up to about four months after the end of infection by Zika.
"We focused on IgG throughout 2017, and this year, we're focusing on the development of a methodology to detect IgM," Oliveira said.
Her group will be assisted by ICB-USP researchers Edison Luiz Durigon and Luiz Carlos Ferreira, as well as Butantan Institute researchers Roxane Piazza and Viviane Botosso.
Although the time taken to obtain a result must be further reduced, the low-cost part of the project's goal has been achieved. Oliveira estimates the cost of the assay to detect IgG at about BRL 10-12 per patient (now about USD 3.00-3.70).
Inovatech has also completed another important step toward bringing its product to market: it has won a Manufacturing Best Practice (BPF) certificate from ANVISA, the national health surveillance agency. The first batches for experimental use are already being produced.