Long QT syndrome—revealed
Long QT syndrome is a rare disorder that can lead to dangerous heart rhythms, fainting and sudden cardiac arrest.
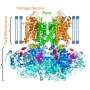
Inherited and potentially lethal forms of the disorder affect the structure and function of the KCNQ1 potassium channel, which transmits electrical signals crucial for maintaining regular heart rhythm.
Now Charles Sanders, Ph.D., and colleagues have used sophisticated cell biological and structural techniques, including NMR spectroscopy, to "classify" mutations according to how structural abnormalities affect protein trafficking and function.
Their findings, reported in the journal Science Advances, could lead to personalized treatment of long QT syndrome in cases where the specific genetic variation underlying loss of function can be determined.
Mutation-induced destabilization of protein folding appears to be the most common mechanism for channel defects causing long QT syndrome, the researchers concluded.
This work also showed that five mutations causing defective folding of the KCNQ1 protein were found in the S0 segment of the voltage sensor domain, highlighting the importance of this previously little-noticed segment in channel function.
More information: Hui Huang et al. Mechanisms of KCNQ1 channel dysfunction in long QT syndrome involving voltage sensor domain mutations, Science Advances (2018). DOI: 10.1126/sciadv.aar2631