Treating triple negative breast cancer by targeting pair of receptors
Triple negative breast cancer (TNBC) is a particularly aggressive form of breast cancer that lacks known targets for therapies, making it difficult to treat. However, a new study by a group of researchers and physician-scientists, led by Baylor College of Medicine, has identified a novel treatment strategy that may be effective for TNBC patients. The study appears in Nature Medicine.
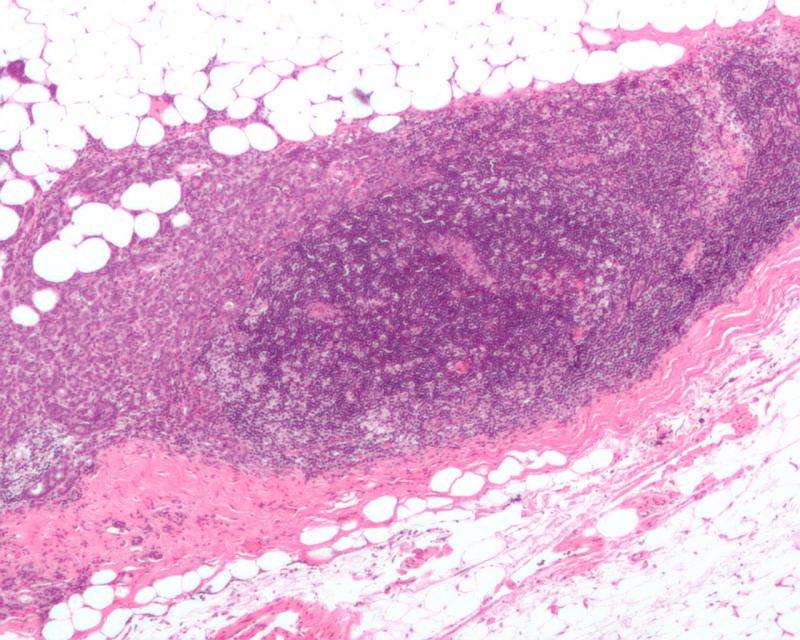
Many types of cancers are caused by receptors that promote the growth of cancers. But the receptors driving some cancer types like TNBC have remained elusive to scientists, until now. In the current study, scientists at Baylor have discovered a pair of receptors that cooperate to drive this deadly disease. Therapies inhibiting these receptors in combination can dramatically shrink TNBCs, including those cancers that are resistant to standard chemotherapy.
Previously, this research team had discovered a gene called PTPN12 that prevents TNBC from occurring. PTPN12 is frequently inactivated in TNBC patients, thus leading to development of the cancer. In the current study, investigators discovered how PTPN12 works to prevent cancer by turning off at least two cancer-promoting receptors. When PTPN12 is inactivated, these receptors become stuck in the "on position" and drive the cancer to grow. This finding led investigators to test the idea that inhibiting both of these key receptors in combination could kill TNBCs.
"This study could have a major impact on our understanding and treatment of triple negative breast cancer, and has prompted two clinical trials," said Dr. Trey Westbrook, director of the Therapeutic Innovation Center at Baylor and McNair Scholar of cancer research. "The first trial testing the effect of inhibiting these receptors was unsuccessful because the two drugs, when used together, were not tolerated by patients. However, a new, single drug has been developed that hits the right combination of receptors, but has much fewer off-targets and side effects, meaning the drug is far more precise in inhibiting these cancer-causing receptors while sparing normal tissues."
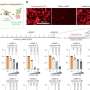
To test the efficacy of inhibiting these receptors in TNBCs, tumors from 14 different patients were grown in mice. While these cancers did not respond to standard chemotherapy, many of them shrank in response to this new therapeutic strategy.
"In TNBCs from half of the patients, we saw tumor regression. This finding could have major clinical implications," said Westbrook, who also is a professor of molecular and human genetics and biochemistry and molecular biology at Baylor. "These results provide a molecular mechanism to match this new therapeutic approach with patients most likely to respond."
The Lester and Sue Smith Breast Center, part of the Dan L Duncan Comprehensive Cancer Center at Baylor, is working through the approval processes to begin enrolling TNBC patients in a clinical trial to test this therapeutic strategy.
"This groundbreaking work offers the most promising prospect of targeted therapy for triple negative breast cancer. We are very excited about exploring this promise in patients with our planned clinical trial," said Dr. Mothaffar Rimawi, associate professor, medical director and clinical research director in the Lester and Sue Smith Breast Center at Baylor.
Dr. Matthew Ellis, professor and director of the Lester and Sue Smith Breast Center and McNair Scholar at Baylor said, "While various approaches to categorizing TNBC have been published, the only ones that matter are those that have the potential to drive therapeutic decisions. This work further solidifies the hypothesis that despite the TNBC label, a substantial number of these aggressive tumors have activated receptor kinases that can be successfully targeted."
In the future, the research team is hopeful that this same therapeutic approach can be applied to many different types of cancers.
More information: Amritha Nair et al. Combinatorial inhibition of PTPN12-regulated receptors leads to a broadly effective therapeutic strategy in triple-negative breast cancer, Nature Medicine (2018). DOI: 10.1038/nm.4507