Can a simple blood test rule out lung cancer?
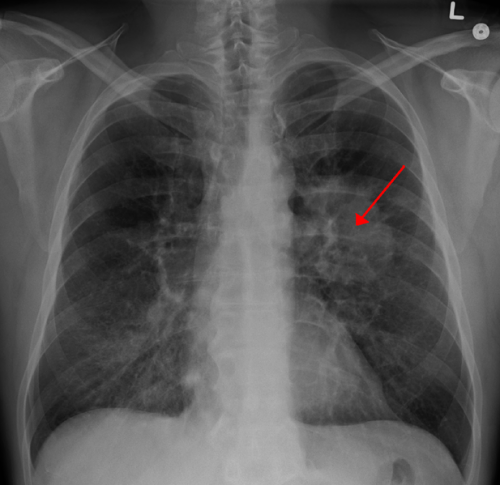
Every year, health care providers in the United States discover more than 1.6 million lung nodules in patients. Many are "incidentally detected," meaning they are found during evaluation for an unrelated cause (for example, a chest X-ray after a fall). Although 75 to 85 percent of these incidentally detected nodules turn out to be benign, they can pose a diagnostic dilemma for providers.
Patients with high-risk nodules may require more invasive testing such as biopsy or even surgery to remove the nodule. However, when there's a low-to-moderate probability of cancer—anywhere from 5 to 65 percent—providers may debate who should be monitored with serial PET or CT scans and who should undergo potentially complex diagnostic tests.
Recently, researchers at the Medical University of South Carolina (MUSC) participated in a multicenter clinical trial to evaluate the accuracy of a blood test, or "biomarker," that measures the levels of two proteins in a patient's plasma, LG3BP and C163A, integrated with clinical predictors of cancer, such as age, size of the nodule and other nodule characteristics. These proteins are common predictors of lung cancer. In the study, the biomarker was 98 percent effective at distinguishing benign from malignant nodules.
Gerard A. Silvestri, M.D., M.S., a lung cancer pulmonologist at the MUSC Hollings Cancer Center who holds the George C. and Margaret M. Hillenbrand Endowed Chair in thoracic oncology, led the study. Results were reported in an article published online in the journal Chest on March 1, 2018.
The biomarker works like this: If a patient has less than a 50 percent chance of having cancer and the test result is negative for the integrated classifier, it's likely not cancer. The biomarker can offer the provider confidence in a diagnosis and treatment plan.
"It serves as a 'rule out' test for those with low-to-moderate risk," says Silvestri. "The biomarker is a tool to help calculate the general risk of cancer and present a patient with recommendations and options. It can push people out of indeterminate risk and into low-risk—without having to undergo invasive and potential risky procedures."
Biopsies and surgeries can be complicated in an organ as delicate as the lung.
"Think of your lung as a two-liter bottle of soda, and the nodule as a pea in the center of it," explains Silvestri. "During a biopsy, for example, the lung could collapse and need a tube to expand it. Our goals for this biomarker are to help calculate the risk of cancer, present the patient with options and recommendations and avoid subjecting patients with benign disease to expensive, unnecessary and intrusive procedures."
Even if biomarker results are negative, patients will need ongoing CT scans to monitor a lung nodule. "A low-risk tumor will be followed with serial imaging. After two years of CT scans being performed periodically and without evidence of growth, we can say it's benign," Silvestri says.
This research is part of the Pulmonary Nodule Plasma Proteomic Classifier (PANOPTIC) study, a clinical trial of 685 patients 40 years old or older, with newly discovered lung nodules 8 to 30 millimeters in diameter as shown on a recent (fewer than 60 days old) CT scan.
If the biomarker's results in the PANOPTIC study had been used to direct care (they were not), 40 percent fewer procedures would have been performed on patients with benign nodules. The next step in bringing this test to the clinic would be to conduct a "clinical utility" study to show how using this biomarker might affect physician and patient behavior.
"Anything to provide physicians with more confidence in how they would manage patients would be helpful," Silvestri says. "The goal of this research, and other research like this, is to quickly evaluate and treat patients with cancerous nodules, while never exposing patients who don't have cancer to invasive costly and sometimes risky procedures."
More information: Gerard A. Silvestri et al, Assessment of Plasma Proteomics Biomarker's Ability to Distinguish Benign From Malignant Lung Nodules, Chest (2018). DOI: 10.1016/j.chest.2018.02.012