New regulator of neuron formation identified
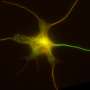
The protein NEK7 regulates neuron formation, as it is required for dendrite growth and branching, as well as the formation and shaping of dendritic spines. These are the main conclusions of a study published in Nature Communications and led by Jens Lüders at the Institute for Research in Biomedicine (IRB Barcelona), in collaboration with colleagues at the University of Barcelona and the Molecular Biology Institute of Barcelona (IBMB-CSIC).
It is firmly established that during cell division, NEK7 regulates microtubules and centrosomes—structures that separate chromosomes during mitosis. However, a function for this gene in neurons has never been reported. Microtubules are tiny filaments that shrink, lengthen, cluster and bend, depending on cell requirements. They are involved in cell mobility, cell division, and intracellular transport, among other functions.
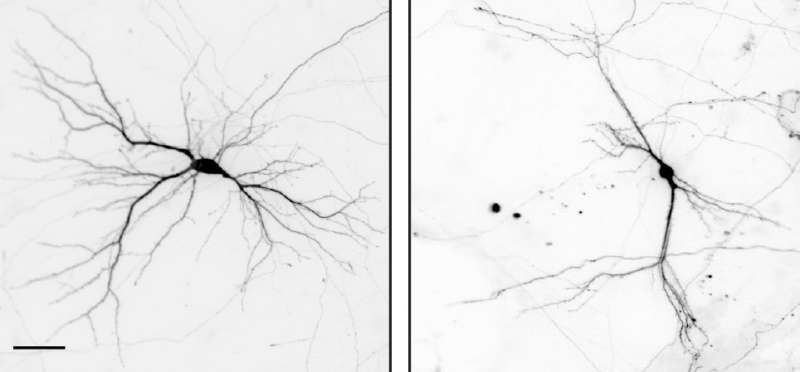
The scientists have used in vitro and in vivo models to show that NEK7 is important for the correct formation of neurons in the hippocampus, a region in the brain involved mainly in the formation of memory. When researchers reduced the levels of NEK7, neurons did not form dendrites correctly. The dendrites became shorter and displayed fewer and improperly formed synaptic structures. The consequences that derive from the malformation of hippocampal neurons remain to be determined, but NEK7 deficiency results in a complex phenotype in mice, thereby suggesting that NEK7 has broader roles, potentially also in other brain regions.
Not just mitosis
Mitosis has been extensively studied, and most microtubule regulators have been identified. This knowledge led to the development of drugs that target microtubules and microtubule-regulating proteins to fight cancer. However, it is often overlooked that, microtubules also have other important functions in post-mitotic differentiated cells such as neurons.
Mitotic cells and neurons may indeed share more microtubule regulators than initially thought. By performing a genome-wide microarray analysis on differentiating neurons in culture, the researchers have identified so-called "mitotic microtubule regulators" that are strongly upregulated in neurons as they differentiate and thus presumably have key roles during this process. "It is of great importance to explore this premise, since not only will it lead to a better understanding of the function of the microtubule network in neurons but it might also help to predict cancer drug side effects," says Francisco Freixo, former "la Caixa" Ph.D. student at IRB Barcelona and first author of the study.
In neurons, for instance, microtubules help to define the shape of these cells and also serve as tracks along which many organelles and neurotransmitters are transported over long distances. "It is not surprising that some chemotherapeutic agents that affect microtubule functions in mitotic cells also have severe side effects in the nervous system," explain Jens Lüders, head of the Microtubule Organization laboratory.
Microtubules determine neuron function and survival. However, to date, little is known about the regulators of the microtubule cytoskeleton in neurons. "In addition to understanding the potential side effects of the chemotherapeutic agents used to target mitotic cells, it is of paramount importance to have a better and more comprehensive knowledge of the identity of microtubule regulators in neurons, what they do, when and how they operate, and what happens when they are not present or when they are misregulated," conclude the researchers.
Mutations in genes encoding microtubule cytoskeleton regulators are associated with neurodegenerative diseases and neurodevelopmental disorders. Examples include TAU, MAP1 and MAP2 associated with Alzheimer Disease, Doublecortin related with brain developmental diseases, and Spastin related to neuronal dysfunction. Future work is likely to reveal more so-called mitotic microtubule regulators that have important roles in the brain.
More information: Francisco Freixo et al. NEK7 regulates dendrite morphogenesis in neurons via Eg5-dependent microtubule stabilization, Nature Communications (2018). DOI: 10.1038/s41467-018-04706-7