Toward a better understanding of Parkinson's disease
A new study, published today in Nature Structural and Molecular Biology, moves researchers closer to understanding one of the crucial proteins involved in Parkinson's disease.
Parkinson's disease is the second most common neurodegenerative disease and commonly affects patients over 60 years of age. It is caused by the degeneration of neurons that are located in the mid-brain and are involved in the dopamine system. One of the causes of the disease is the failure to clear away the damaged mitochondria within these neurons. Mitochondria are the powerhouse of the cells, creating energy for the cell.
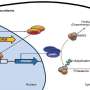
Mutations in a protein, parkin, are responsible for a genetic form of the disease. Now, by using powerful beams of x-rays, a McGill-based research team, led by Kalle Gehring, has been able to visualize the steps of activation of this crucial protein. With funding from the Michael J Fox Foundation and the Canadian Institutes of Health Research, the team is learning how parkin is regulated within cells. This research offers hope for using parkin as a target for therapeutic agents to slow or prevent the progression of Parkinson's disease.
More information: Véronique Sauvé et al. Mechanism of parkin activation by phosphorylation, Nature Structural & Molecular Biology (2018). DOI: 10.1038/s41594-018-0088-7