Watching neurons in action
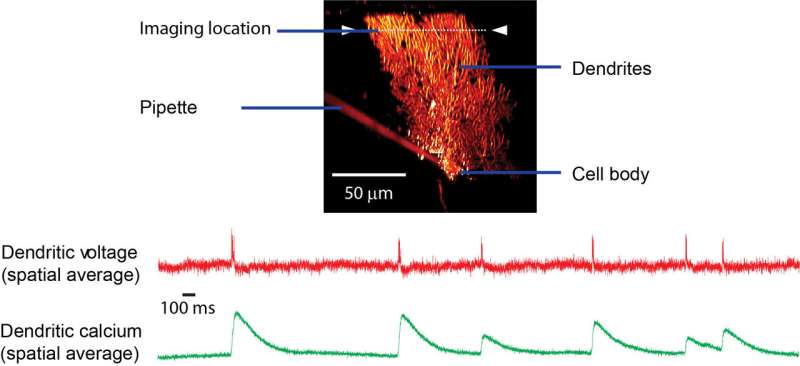
Capturing the activity of neurons in the form of electric impulses isn't easy. Information processing happens on different time scales and involves quick changes in voltage, ion concentrations and multiple signaling molecules. In a new study published in the journal Nature Communications, scientists from the Optical Neuroimaging Unit at the Okinawa Institute of Science and Technology Graduate University (OIST), report a newly developed imaging technique that maps electrical activity in single neurons in animals that are conscious. This was previously a challenge due to the delicate structure of neurons and the fast changes in voltage.
In most studies on how neurons communicate, scientists record electrical signals from neurons within a thin slice of brain tissue maintained in a dish. In such experiments, neurons can be observed in isolation, without almost any inputs from other neurons. "From the brain slice technique, we learnt a lot of how neurons work, but it fails to give us a complete picture of the way neurons function in living animals where each neuron is connected with thousands of other neurons," explains Prof. Bernd Kuhn, the principal investigator of the study. "Now we can overcome this issue."
Prof. Kuhn and Dr. Christopher Roome, a postdoctoral researcher at OIST, have used the new technique to record the activity of Purkinje neurons, which are found in the cerebellum—a part of the brain mainly involved in motor coordination. Each of these Purkinje neurons receives inputs from about 20,000 other neurons in a mouse, and, for comparison, around 200,000 in humans.
The secret to the scientists' success was an orange dye called ANNINE, which was originally developed at the Max Planck Institute of Biochemistry, Germany. Prof. Kuhn was involved in its development 20 years ago and has been working ever since on optimizing it for voltage imaging. The dye and the accompanying imaging technique now allow researchers to detect even the minutest change in voltage inside a neuron.
For the development of this new technique, the highest priority was animal wellbeing, for ethical and scientific reasons. The scientists began by making a very small incision in the skull to access Purkinje neurons in a mouse's brain and genetically modified them to produce a protein named GCaMP, a green dye that tracks calcium ion concentrations within a neuron. Dr. Roome developed a special technique to close the incision previously made, while providing a window to observe neuron activity and still allowing access to the neurons through a port. This port made it possible for the scientists to inject the voltage-sensitive ANNINE dye, one cell at a time, using a micro-pipette. The neurons were then observed using a two-photon microscope.
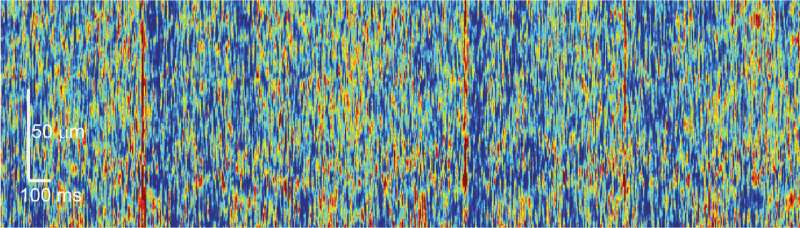
This double labeling of neurons with ANNINE and GCaMP allows mapping their activity through simultaneous imaging of voltage and calcium concentrations. When a neuron is activated, the voltage inside it changes within a thousandth of a second. It also causes the calcium levels inside the cell to fluctuate, but they change around 10 to 100 times more slowly. So by mapping the voltage changes, the researchers were able to detect different types of signals than those detected by measuring calcium ion concentrations, that occur within a single neuron. Based on this voltage mapping technique, Prof. Kuhn and Dr. Roome estimated each Purkinje neuron to receive around 10000 signals per second from other neurons. Image 2 above depicts the signals as red dots and streaks.
"This is a study of many firsts," says Prof. Kuhn. Not only is it the first time a voltage-sensitive dye has been used to detect and map the activity of a single neuron in a conscious animal, it is also the first simultaneous imaging of electrical activity and calcium ion concentration. "The optical recordings of this study are the most complete observation of how a single neuron works in a conscious animal to date," says Prof. Kuhn. The scientists believe this new technique will allow neuroscientists to learn how neurons—the basic building blocks of our brain—function in an awake and responsive animal.
More information: Christopher J. Roome et al, Simultaneous dendritic voltage and calcium imaging and somatic recording from Purkinje neurons in awake mice, Nature Communications (2018). DOI: 10.1038/s41467-018-05900-3