Guardians of the Genome

For many, breast cancer is more than just a disease – it's personal. One in eight women will be diagnosed with breast cancer at some point in their lives. But through new discoveries at the genetic level, the personal nature of cancer will eventually be what helps to beat it.
DNA repair – key defense against mutations
A healthy individual has a system of checks and balances that curtail cells' irregular growth. But changes occurring during a person's lifetime, including inherited changes and those induced by environmental exposure, alter the body's normal blueprint and cause cancer.
"One key to understanding cancer lies in the stability of the genome," says Distinguished Professor Wolf-Dietrich Heyer, Department of Microbiology and Molecular Genetics chair and co-leader of the Molecular Oncology Program at the UC Davis Comprehensive Cancer Center.
Many genes, as part of regular maintenance within the body, are responsible for repairing damaged DNA. Through a process called homologous recombination, information from healthy DNA molecules is used as a template to heal broken DNA strands.
"Recombination works like accessing a backup version when a file on your computer is compromised," Heyer says. "Referencing this genetic backup copy enables high-fidelity DNA repair."
Proteins involved in recombination are constantly directing cellular repair, serving as genetic guardians to reinforce a healthy blueprint for the body. It is estimated that each day, every cell experiences tens of thousands of damaging events that require DNA repair. These guardians stay busy and are constantly on the alert.
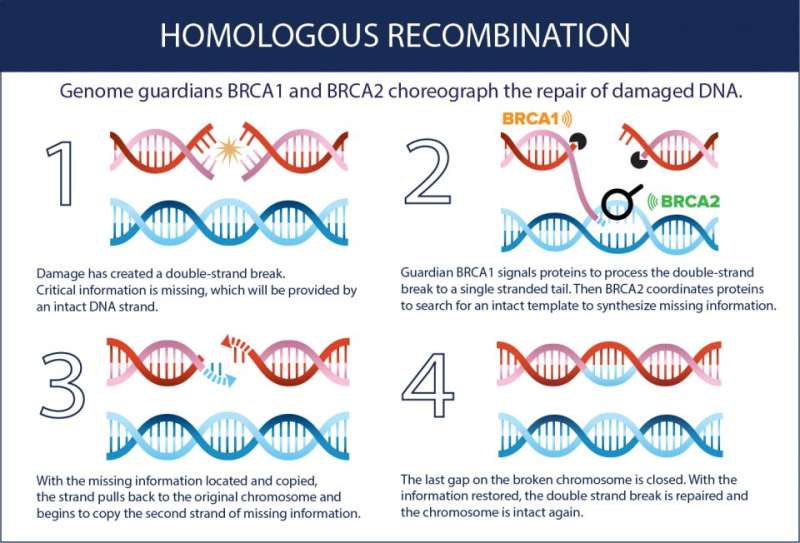
While the guardians are tasked with repairing DNA damage, they can also become damaged themselves. When they become altered or damaged, genetic changes, called mutations, accumulate at an alarming pace. Mutations in two very different guardian genes, BRCA1 and BRCA2, lead to a significantly elevated cancer risk.
According to the National Cancer Institute, 55 to 65 percent of women who inherit the BRCA1 mutation will develop breast cancer by age 70, while 39 percent will develop ovarian cancer. Together, deficiency mutations in these two genes constitute the highest risk factor for familial breast and ovarian cancer.
There are many types of risks that can increase the likelihood of these cancers in women. Fortunately, the consistency of BRCA1 and BRCA2 deficiency mutations in cancer patients creates an opening that UC Davis researchers have seized to gain understanding about their roles in preventing and treating cancer.
Understanding cancer, one building block at a time
Due to the complexity of the proteins produced by the genes, uncovering the characteristics of BRCA1 and BRCA2 has been a decades-long process. BRCA2 contains 3,418 amino acids, making it roughly 10 times larger than an average protein, Heyer says. The larger the protein, the more challenging it is to understand. And because genes mutate in unpredictable ways, they also end up producing many different variations of proteins.
"There may be only a single person on the planet who has a particular variant," says Heyer, "and with a protein of this size, it is very difficult to predict what impact that change has, leaving patients and doctors in the dark."

Insights into the nature of BRCA1 and BRCA2 began with identifying similar protein activities in bacteria and yeast, simpler organisms that share a common ancestor with humans.
Professor Stephen Kowalczykowski, Department of Microbiology and Molecular Genetics, has devoted his research career to understanding the mechanisms of the proteins responsible for recombination.
"Without the knowledge derived from bacteria, we would know that BRCA1 and BRCA2 mutations exist, but wouldn't know what they do nor how to treat them," Kowalczykowski says. "What's been interesting and exciting about this is the ability to take what has been a very abstract system study in bacteria and apply what we learned there. It educates us about the processes in humans."
Through long-term and painstakingly detailed research, Kowalczykowski has sought to replicate the cellular processes of DNA repair in a lab setting. With a sophisticated microscope, Kowalczykowski views live interactions of individual proteins functioning on single molecules of DNA.
"There was never one defining moment," says Kowalczykowski. "Every five or 10 years you would come to the realization that something in bacteria is identical in humans. Once you realize the universality of the underlying process you realize that everything you learned in bacteria applied to humans, but those human cells are just more complicated."
Personalized medicine, the future of cancer therapies
Heyer, Kowalczykowski and dozens of other UC Davis researchers' discoveries are creating the framework for the future of cancer therapies. By describing the behaviors of cancer-related genes such as BRCA1 and BRCA2, it will become possible to create specific profiles to target tumor growth. One day, physicians will be able to prescribe a customized treatment plan for each patient, based on their genetic profile.
"Many tumors, once they progress, accumulate sporadic mutations," Kowalczykowski says. "If you knew what the mutations were in a particular patient's cancer you could personalize a treatment. It's all a knowledge-based form of decision-making. Compared to 10 to 20 years ago, it's easier to find the base of mutation; it's easy to screen once you have descriptions of mutation and DNA repair genes. How we treat these mutations, that's the next generation of discovery."
The main approaches of cancer therapy are surgery, radiation and chemotherapy, often associated with severe side effects. Personalized therapies based on the individual genetic profile provide an alternative, or supplement, to chemotherapy and radiation treatments, which indiscriminately kill healthy cells and further damage the guardian genes' ability to repair DNA.
These targeted therapies can seek out cancerous cells based on the specific, individual genetic makeup. So while DNA repair defects caused by mutations in BRCA1 and BRCA2 predispose individuals to cancer, they also make tumors uniquely susceptible to certain targeted treatments that have little effect on normal cells.
The goal is to develop personalized treatments based on an individual's genetic profile that can target cancers while leaving healthy cells intact. Through collaboration with the UC Davis Comprehensive Cancer Center, researchers and bedside clinicians are working together to bring cancer treatment full-circle to improve patient care.

A patient's genetic makeup and cancer type can also provide access to early-stage clinical trials for new medications. Methodologies of personalized care continue to advance, and one day, enough information may exist to adapt any cancer treatment to an individual's unique genome.
"To have so much biological knowledge that each person can be treated in a personalized way, that is the long-term strategy," says Heyer. "Our effort is to establish the basic science and help translate these insights into the cancer clinic."
While DNA repair might not be a silver bullet for all cancers, it is a leading methodology that aims to address the shortcomings of traditional treatments. Personalized medicine approaches based on an individual's genetic profile are already saving lives by revealing predispositions to certain types of cancer before tumors begin to form.
As UC Davis researchers continue to investigate the genetic processes underlying cancer, therapies will shift to become more personalized and patient-specific. New methods will spur increased collaboration between researchers and physicians, improving a working model of feedback that will accelerate cancer treatments and help save lives. And as anyone who has been affected by cancer knows, a personal approach makes all the difference.



















