Certain blood pressure drugs linked to increased risk of lung cancer
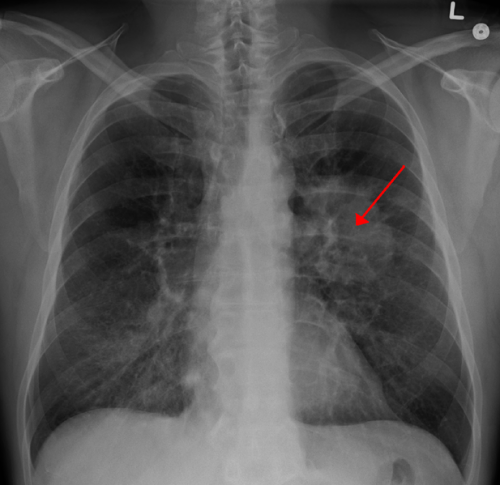
Use of angiotensin converting enzyme inhibitor drugs (ACEIs) to lower blood pressure is associated with an increased risk of lung cancer compared with use of another group of blood pressure drugs called angiotensin receptor blockers (ARBs), finds a study in The BMJ today.
The risk is particularly elevated among people using ACEIs for more than five years, say the researchers.
Although the risk for individual patients is modest, ACEIs are widely prescribed, so these small relative effects could translate into large absolute numbers of patients at risk for lung cancer, say the researchers.
ACEIs are effective drugs used to treat high blood pressure (hypertension). Evidence suggests that ACEIs may increase the risk of lung cancer through the build-up of protein-like chemicals called bradykinin and substance P in the lung. These chemicals have been found on lung cancer tissue, and bradykinin may directly stimulate the growth of lung cancer.
However, previous observational studies examining this association are limited and report inconsistent results.
To better understand this possible association, researchers led by Professor Laurent Azoulay at McGill University in Canada analysed UK primary care records for nearly one million patients who started taking a new antihypertensive drug between 1995 and 2015.
Patients were at least 18 years of age, with no previous cancer, and were followed up for an average of 6.4 years, during which time 7,952 cases of lung cancer were identified (a rate of 1.3 per 1000 person years).
After taking account of factors that could potentially influence the findings, including age, sex, weight (BMI), smoking status, alcohol related disorders, and history of lung diseases, use of ACEIs was associated with an overall 14% increased risk of lung cancer compared with ARBs (1.6 v 1.2 cases per 1000 person years).
Associations were evident after five years of use and increased with longer durations of use, particularly in patients who used ACEIs for more than 10 years (31% increased risk).
Although the magnitudes of the observed estimates are modest, the researchers point out that ACEIs are one of the most widely prescribed drug classes, "so these small relative effects could translate into large absolute numbers of patients at risk." As such, they say these findings "should be replicated in other settings, particularly among patients exposed for longer durations."
This is an observational study, so no firm conclusions can be drawn about cause and effect, and the researchers cannot rule out the possibility that other unmeasured factors, such as socioeconomic differences, diet, and family history of lung cancer, may have affected the results.
However, this is the largest study to assess this specific association, and findings remained consistent after further analyses to test the strength of the results.
As such, the researchers conclude that the use of ACEIs is associated with an increased risk of lung cancer, and they call for "additional studies, with long term follow-up, to investigate the effects of these drugs on incidence of lung cancer."
In a linked editorial, Associate Professor Deirdre Cronin Fenton from the Department of Clinical Epidemiology at Aarhus University in Denmark, says although a 14% relative increase in lung cancer incidence might not translate to a large absolute risk, "the findings are important given the considerable use of ACEIs worldwide."
Nonetheless, in an individual patient, concerns about the long term risk of lung cancer "should be balanced against gains in life expectancy associated with use of ACEIs," she writes. "Further studies with long term follow-up are now needed to enhance the scientific evidence on the long term safety of these drugs," she concludes.
More information: Angiotensin converting enzyme inhibitors and risk of lung cancer: population based cohort study, BMJ (2018). www.bmj.com/content/363/bmj.k4209
Editorial: Angiotensin converting enzyme inhibitors and lung cancer, BMJ (2018). www.bmj.com/content/363/bmj.k4337