Machine learning uncovers dementia subtypes with implication for drug trials
Machine learning could help to find new treatments for dementia, according to researchers at UCL.
A new algorithm that can automatically disentangle different patterns of progression in patients with a range of different dementias, including Alzheimer's disease, will enable individuals to be identified that may respond best to different treatments.
For the paper, published in Nature Communications, researchers devised and applied a new algorithm called SuStaIn (Subtype and Stage Inference) to routinely acquired MRI scans from patients with dementia.
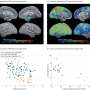
The algorithm was able to identify three separate subtypes of Alzheimer's disease, which broadly match those observed in post-mortems of brain tissue, and several different subtypes of frontotemporal dementia. Critically, however, this subtyping could be done in life, using brain scanning, and very early in the disease process.
Being able to identify the subtypes early on in the disease process and using non-invasive MRI scanning means there is a better chance of identifying the best treatment for individuals.
Professor Daniel Alexander (UCL Centre for Medical Image Computing) said: "This new algorithm has the unique ability to reveal groups of patients with different variants of disease. One key reason for the failure of drug trials in Alzheimer's disease is the broad mixture of very different patients they test; a treatment with a strong effect on a particular subgroup of patients may show no overall effect on the full population so fail the drug trial.
"SuStaIn provides a way to show treatment effects on distinct subgroups, potentially expediting treatments to market."
Classic studies of dementia in individuals take a single measurement based on their symptoms, and aren't clear on what stage the disease is in.
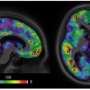
SuStaIn uses medical imaging, which allows doctors to see how the disease is progressing, look at the specific locations of protein build up within the brain, and deduce which parts are degenerating.
Dr. Alexandra Young (UCL Centre for Medical Image Computing) said: "Individuals might present with similar symptoms to each other, but using SuStaIn we can find that they belong to different subgroups. This allows us to predict more accurately how their disease will progress and diagnose it earlier."
Professor Jonathan Schott (UCL Institute of Neurology) said: "Understanding how different diseases evolve over time is critical if we are to design rational treatment trials and inform patients about prognosis.
"This is a major challenge for diseases that evolve over years, if not decades, and where there may be substantial differences in the underlying pathology, and pattern and rates of progression between patients.
"This work shows that it is possible to tease out different disease patterns – some hitherto unknown – from single MRI scans taken from patients with a range of different dementias. As well as providing new insights into dementia, this work demonstrates the huge potential of SuStaIn to delineate disease subtypes in a range of other medical contexts."
The team are now looking for ways to apply the algorithm to other diseases, having recently presented it at the European Respiratory Society Conference on chronic obstructive pulmonary disease (COPD).
More information: Uncovering the heterogeneity and temporal complexity of neurodegenerative diseases with Subtype and Stage Inference, Nature Communications (2018). DOI: 10.1038/s41467-018-05892-0