Diabetes-inducing blood vessel damage could be prevented by a growth factor, study finds
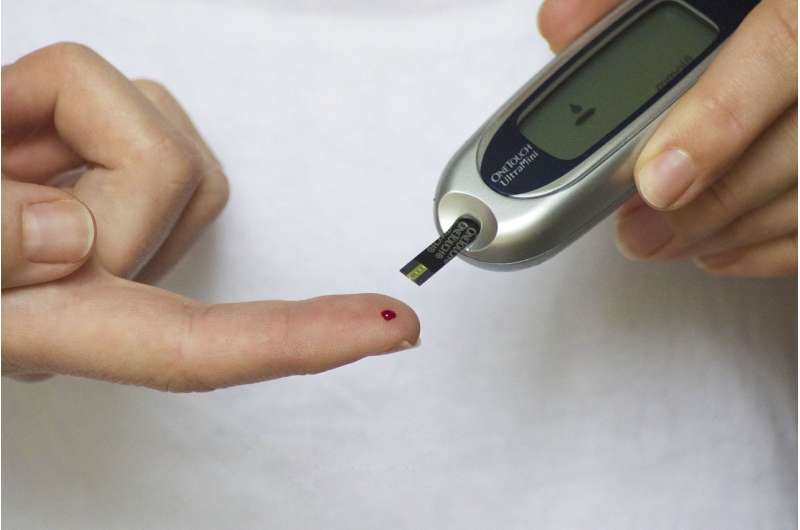
Scientists have identified a growth factor found in the kidneys that could minimise the diabetes-inducing effects of blood vessel damage.
The findings come in the wake of recent data from Diabetes UK which revealed that almost 4.6 million people nationwide have either type 1 or type 2 diabetes. Coupled with the recent NHS announcement that diabetes prescriptions now cost the UK £1bn a year, this latest research offers fresh hope for preventing an increasingly prevalent disease.
The research, led by experts in renal disease and translational health sciences from the University of Bristol and funded by the British Heart Foundation, focused on a group of growth factors categorised as VEGFs, or vascular endothelial growth factors. These are growth factors produced by cells that stimulate the formation of blood vessels.
In health, the subtype VEGFA plays a vital role in the kidney ensuring that blood vessels can filter and so allow the kidneys to effectively remove waste products and excess fluid from the body, while retaining other vital substances. However, too much VEGFA can damage blood vessels, causing them to leak substances that should be retained, which reduces their functionality and so exacerbates disease.
"We wanted to know whether we could protect the kidneys by using another growth factor from the same family of proteins to balance this effect," explains Dr. Rebecca Foster, from Bristol University's Translational Health Sciences, part of the Bristol Medical School.
Researchers were able to express this other growth factor subtype (VEGFC) in the kidneys of a mouse model as diabetes developed. The mice expressing VEGFC were protected from the development of diabetic kidney disease.
The study found that the growth factor VEGFC can counterbalance the effects of VEGFA by protecting the blood vessels. Pending further research, these findings could ultimately prove instrumental in how clinicians may protect those with diabetes from the development of diabetic kidney disease
The findings are published today [20 December] in the journal Diabetes.
"We now have a better understanding of how to protect filtering blood vessels in the kidney," added Dr. Foster. "Blood vessel damage is an early event in the development of diabetic kidney disease. By activating the VEGFC signalling pathway, we can potentially prevent the development of diabetic kidney disease.
"The next step is to identify the key part of the VEGFC pathway that leads to reduced blood vessel leakiness. Then we need to demonstrate how this aspect could be selectively activated to protect people from diabetic kidney disease."
Professor Metin Avkiran, Associate Medical Director from the British Heart Foundation, which funded the research, said: "In the UK, around 4.6 million people are living with diabetes. The damage this condition does to our blood vessels can cause diabetic kidney disease, as well as a whole range of heart and circulatory diseases. A third of adults with diabetes will die from one of these conditions.
"Understanding how we can protect blood vessels in people with diabetes could help us to find ways to prevent and treat many conditions, including coronary heart disease, stroke and vascular dementia."
More information: Karen L. Onions et al. VEGFC Reduces Glomerular Albumin Permeability and Protects Against Alterations in VEGF Receptor Expression in Diabetic Nephropathy, Diabetes (2018). DOI: 10.2337/db18-0045