Killing tumours by targeting their viral DNA
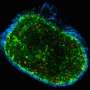
Epstein-Barr virus infects more than 95 percent of people, usually without symptoms. But sometimes its persistence in cells can lead to tumour formation. Now, researchers from Hong Kong and the UK have developed a fluorescing, molecular-sized probe, called L2P4, which can inhibit Epstein-Barr-related tumour growth while allowing researchers to see the targeted tumour cells.
A team led by chemist Gary Ka-leung Wong from Hong Kong Baptist University developed the probe, which is formed of a nucleus-penetrating peptide linked to a fluorescent chemical compound. L2P4 targets a viral protein, called EBNA1, which is crucial for maintaining the Epstein-Barr virus (EBV) genome inside tumours.
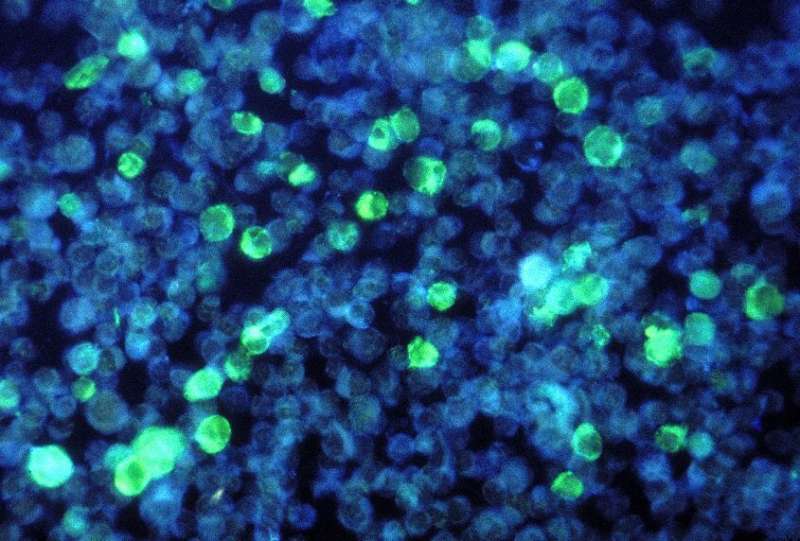
The team tested the probe in EBV-positive tumour cells cultured in petri dishes and in EBV-positive tumours grafted into mice. They found that the peptide was only minimally toxic to cells that did not contain the virus, but that it became more toxic to EBV-positive tumour cells the higher the dose. Also, injecting L2P4 into EBV-positive tumours grafted into mice drastically reduced tumour size by 93 percent.
L2P4 works by disrupting the function of the viral protein EBNA1, interfering with the replication of EBV's genome in tumour cells, as well as reducing tumour cell stability.
But further investigations are needed. The researchers injected L2P4 directly into tumours, but localized tumours are already treated effectively by surgical removal or radiation. Injecting the probe into the body's general circulation could lead to an immune response with a subsequent rapid reduction in its levels, preventing effective delivery to tumour cells.
Still, the probe is a promising target as a future tumour therapy. Its ability to fluoresce in EBV-positive tumour cells also means it could potentially be used in cancer imaging.
The team next plans to map intracellular zinc ions in EBV-infected cells. Zinc is involved in the regulation of EBNA1. The researchers would like to investigate adding a molecule that binds zinc to their probe to enhance its therapeutic effectiveness.
More information: Lijun Jiang et al. EBNA1-targeted probe for the imaging and growth inhibition of tumours associated with the Epstein–Barr virus, Nature Biomedical Engineering (2017). DOI: 10.1038/s41551-017-0042