Breakthrough device lures aggressive brain tumor cells out of the patient
A biomedical tool that tricks aggressive brain tumors such as glioblastoma into migrating into an external container rather than throughout the brain has been designated a "Breakthrough Device" by the U.S. Food and Drug Administration (FDA).
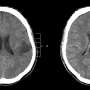
Dubbed the Tumor Monorail, the device mimics the physical properties of the brain's white matter to entice aggressive tumors to migrate toward the exterior of the brain, where the migrating cells can be collected and removed. The purpose of the device is not to destroy the tumor, but to halt its lethal spread, making the disease more of a condition to manage than a death sentence.
Breakthrough designations from the FDA aim to expedite the development and review of drugs, diagnostics and devices aimed at life-threatening or irreversibly debilitating conditions. While the designation does not mean that the device has been approved for clinical use, it does provide a partnership with the FDA that can speed development, assessment and review.
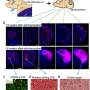
The Tumor Monorail first made headlines in 2014 in an initial exploratory study published in Nature Materials and led by researchers at Georgia Tech and Emory University that showed the concept was effective in rat models. A prototype device successfully played Pied Piper to glioblastoma cells, enticing them to migrate toward a repository filled with a toxic gel. The result was that the tumors' spread slowed and they shrank by more than 90 percent.
"This was the first demonstration that you can engineer migration inside the body and move a tumor from point A to point B by design," said Ravi Bellamkonda, the Vinik Dean of the Pratt School of Engineering at Duke University, who began the research while at Georgia Tech. "It was also the first demonstration of bringing the tumor to your drug rather than your drug going into the brain and killing valuable cells."
The device works by mimicking the physical structures of the brain's white matter where it travels through a narrow opening connecting the left and right hemispheres—a popular growth track for glioblastoma. There are no chemicals or enzymes involved, and there are a wide variety of materials that the device could be made from.
"The tumor monorail device is a true game-changer in how we think about treating brain tumors," said Barun Brahma, a neurosurgeon at Children's Healthcare of Atlanta, who has been a member of the research team since the beginning. "There are many tumors that are considered inoperable due to the location of the tumor or the frailty of the patient. This device affords clinicians the ability to surgically treat these tumors with a minimal approach."
Since the initial study, the results in rats have been successfully reproduced multiple times by the research group, which has also been busy scaling up and redesigning the device for potential human use.
Because the material itself isn't as important as its physical structure, the researchers have swapped a material into their design known to last a long time and be well tolerated by the human body. In its current iteration, the device resembles a long, thin catheter tube with a small reservoir at the end that sits on top of the skull under the scalp. The researchers have also done away with the toxic gel contained in the reservoir to simplify FDA approval.
"What's most important is that the tumor is spreading in a controlled way through our device to a reservoir, and away from the mother tumor, rather than through the healthy brain tissue," said Nassir Mokarram, a research faculty member in the Department of Biomedical Engineering at Duke University and leader of the project. "The toxic gel inside the reservoir appears to only play a secondary role, though additional preclinical studies will help make this clear. Simply by being far away from the mother tumor, the cells are more susceptible to dying anyway, and a neurosurgeon can access the reservoir to empty it when needed."
The researchers are now working to prove the device is safe for human trials—a task that is easier said than done.
"The most exciting part about this designation is that it gives us the opportunity to look at the FDA as a partner rather than a reviewer," said Mokarram. "The task before us is to prove that the device does not pose a risk to human patients, which is an extremely complicated process. With direct access to the FDA reviewers, we can get more efficient, faster feedback on our experimental ideas to make sure we're addressing all of their concerns from the very start."
With the help of the FDA, the research group hopes to solidify their experimental plans within the next several weeks so that they can begin their next study as soon as possible. They are working toward gaining FDA approval for human trials by the end of 2019.
More information: Anjana Jain et al. Guiding intracortical brain tumour cells to an extracortical cytotoxic hydrogel using aligned polymeric nanofibres, Nature Materials (2014). DOI: 10.1038/nmat3878