Researchers develop biomarkers, blood test that shows MS patients in relapse
Dr. Horea Rus is an expert in diagnosing and treating patients with multiple sclerosis, and his research laboratory at the University of Maryland is producing new tools for treating the disease that attacks the central nervous system.
On Tuesday, Rus and former postdoc research fellow Cosmin Tegla were issued U.S. Patent 10,280,465 for developing a biomarker, using Sirtuin 1, RGC-32, FasL, and IL-21 proteins, that indicates if a patient with relapsing-remitting MS is in relapse.
The most common tests for determining if a patient has MS or is in relapse (magnetic resonance imaging, visual evoked potential testing, or analysis of the cerebrospinal fluid) are expensive, can be painful, and may induce negative side effects.
But an estimated 1 million people in the U.S. have MS, including 60,000 military veterans. That's why the doctors, funded by the Department of Veterans Affairs, explored alternatives.
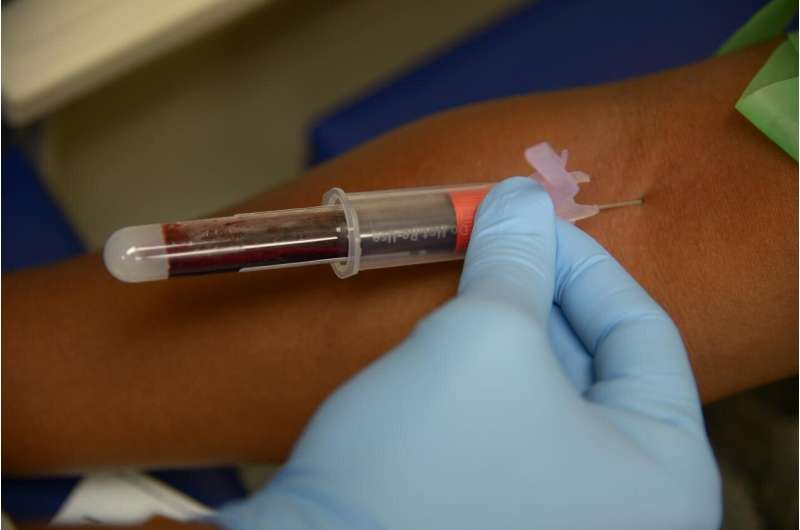
Their research developed the foundation for a fast and accurate diagnostic tool through analysis of the biomarker proteins in a patient's blood.
"The data obtained from the method will indicate whether the subject is undergoing relapse. Based on this information, a physician can determine the best course of new or continuing treatment for the subject, according to the patent.
"Thus, when the method indicates that the subject is undergoing relapse, [a doctor can administer] a therapeutically effective amount of a treatment for relapsing-remitting multiple sclerosis to the subject. The treatment may be, for example, glatiramer acetate. Other suitable treatments for relapsing-remitting multiple sclerosis include, but are not limited to, beta-interferons, teriflunomide, fingolimod, dimethyl fumarate, and natalizumab."
In coordination with the VA Technology Transfer Program, TechLink is helping innovative companies evaluate the technology for the development of new health care products and services.
TechLink is the VA's national partnership intermediary for technology transfer, and for 20 years has been guiding businesses through federal patent licensing at no charge.
Gary Bloomer, a senior technology manager at TechLink, is managing the marketing, assisting in the evaluation, and facilitating licensing of the VA's patent portfolio, which includes helping companies draft the necessary license application and commercialization plan.
"Through technology transfer, qualified businesses can benefit from the VA's investment in research and expand patient care with new capabilities," Bloomer said.