Dupixent approved for chronic rhinosinusitis with nasal polyps

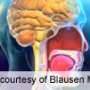
Dupixent (dupilumab) has been approved to treat nasal polyps in adults with chronic rhinosinusitis, the U.S. Food and Drug Administration announced today.
Dupixent, which is administered through injection, was previously approved to treat eczema in patients 12 years and older and as an add-on maintenance treatment for moderate-to-severe eosinophilic asthma or oral corticosteroid-dependent asthma. The recommended dose for adult patients with chronic rhinosinusitis and nasal polyps is 300 mg every other week.
Efficacy and safety trials were conducted in 724 patients aged 18 years or older with chronic rhinosinusitis and nasal polyps. Patients were taking intranasal corticosteroids but were still symptomatic. Researchers found statistically significant reductions in nasal polyp size and nasal congestion with Dupixent compared with placebo. Patients who took Dupixent reported improved ability to smell and required less nasal polyp surgery and oral steroids.
Commonly reported side effects included injection site reactions and arthralgia. Dupixent can cause serious allergic reactions and eye problems including conjunctivitis and keratitis. The manufacturer's prescribing information also lists eosinophilia, insomnia, toothache, and gastritis as possible adverse reactions. The FDA notes that patients should not receive live vaccines when taking Dupixent.
Approval was granted to Regeneron Pharmaceuticals.
More information: More Information
Health News
Copyright © 2019