A case for personalized medicine in the search for answers about cancer
A new cancer is diagnosed every 30 seconds in the United States. Every three minutes, two people in the U.S. die from cancer. Some of the deaths related to the disease arise from the fact that the same drug that helps one person can be detrimental to another with the same type of cancer.
"This is one of the strong cases for personalized medicine," said Nadia Atallah Lanman, a research assistant professor in Purdue University's College of Veterinary Medicine, who manages the Purdue University Collaborative Core for Cancer Bioinformatics (C3B). "We are training the next generation of scientists to be able to move oncological research forward in a data-driven manner."
The C3B is a joint bioinformatics core directed by Jun Wan, an assistant professor at Indiana University, between the Purdue University Center for Cancer Research (PCCR) and the Indiana University Melvin and Bren Simon Cancer Center (IUSCC). These two cancer centers are designated by the National Cancer Institute as top-tier cancer research organizations, with IUSCC recently achieving comprehensive status.
Bioinformatics is a diverse interdisciplinary field, which combines statistics, biology, computer science, mathematics and engineering to analyze and interpret complex biological data sets. The scientists seek to understand basic and applied research questions about the development of cancer and predict which people will respond well to which treatments.
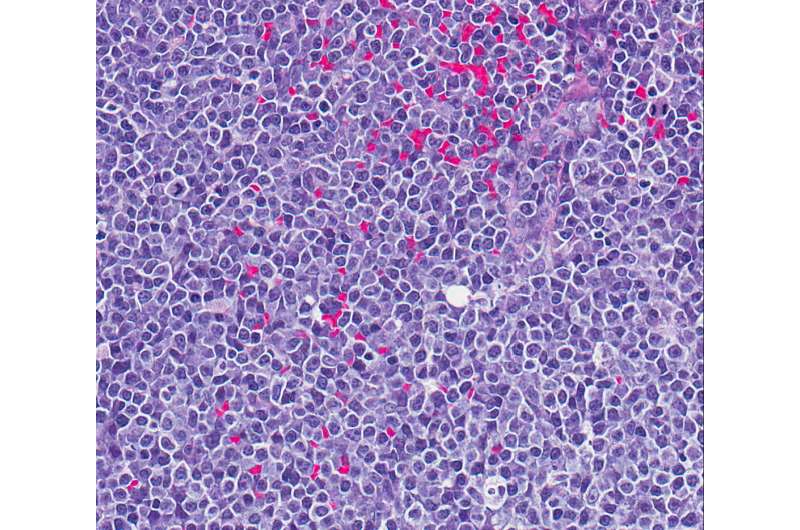
C3B scientists explore diverse biological big data to perform different analyses, such as identifying mutations in the genome or gene expression patterns that may be associated with or cause a specific type of cancer. The C3B collaborated with Purdue faculty members Mike Childress, John Turek and David Nolte on a project that uses pet dogs with naturally occurring diffuse large B-cell lymphoma (DLBCL) as a model for studying chemotherapy resistance in lymphoma patients.
DLBCL is an aggressive form of non-Hodgkin's lymphoma and half of patients diagnosed with the disease will die because of the development of chemotherapy resistance. The project combined gene mutation data, gene expression data and data from a technique, biodynamic imaging (BDI), developed by Nolte and Turek, which performed phenotypic profiling of three-dimensional tissue samples.
Genetic mutations, BDI data and gene expression signatures related to the development of chemotherapy resistance and poor treatment outcome were identified. This study could change the way doctors prescribe treatments for DLBCL, as well as identifying molecules and processes that can be targeted by new chemotherapeutics, which could help patients who show resistance to standard chemotherapy regimens.
"The fact that the two universities are working together to continue building and running the C3B is truly a strength," Lanman said. "These complex problems are not going to all be solved by one researcher. We must work together to develop personalized therapies and to really get to the heart of what causes cancer, and what can be done to prevent cancer."
Wan said, "Our core has a unique bioinformatics team which spreads out over the two campus and has had a great impact already on the research performed at both institutions. The C3B has successfully assisted 107 members from two cancer centers on 306 different projects during the past three to four years."
Tim Ratliff, the Robert Wallace Miller Director of the Purdue University Center for Cancer Research, has worked with C3B bioinformaticians from both IUSCC and PCCR in his research, which focuses on understanding immune regulation and the development of novel approaches for treating urologic cancers such as bladder cancer and prostate cancer through the modulation of anti-cancer immunity.
The Ratliff group, in collaboration with the C3B, has been using single-cell RNA sequencing to probe the heterogeneity in urologic cancers and to identify changes in gene expression that correspond to disease development and severity, as well as to identify targets which could be exploited to develop new treatments for these types of cancers. So far, this project has led to the identification of pathways and potential targets that could enhance treatment of castration resistant prostate cancer, which is currently incurable.