FDA warns of surgical gowns that may not be sterile
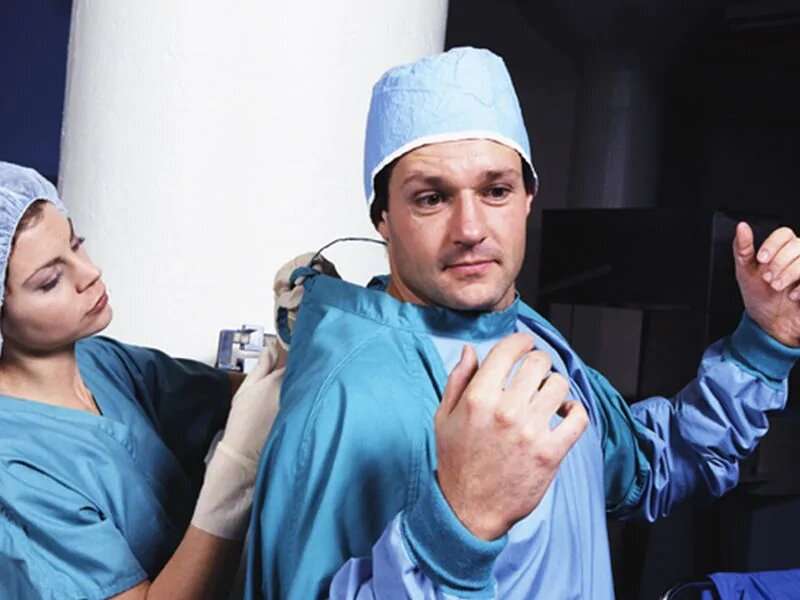
Certain surgical gowns and surgical packs made by Cardinal Health may not be sterile and should not be used, the U.S. Food and Drug Administration said Thursday.
On Jan. 11 and Jan. 15, 2020, the medical device maker alerted its customers about quality issues affecting some of its Level 3 surgical gowns and PreSource procedural packs that contain these gowns.
The FDA said it is working with Cardinal Health to "understand and address the quality issues with these products, including the potential risks to users and patients, which specific product lots are impacted, and the potential impact on the supply chain."
The agency said it's "concerned about possible contamination of the products and agree[s] with the manufacturer's recommendation about not using the affected lots of Level 3 surgical gowns or PreSource procedural packs," adding that there are a number of other FDA-approved surgical gowns that provide Level 3 protection.
The FDA has not been notified of any patient harm because of this issue.
More information: More Information
Copyright © 2020 HealthDay. All rights reserved.