New type of heart valve may be the only replacement a child needs

Current prosthetic heart valves for children with congenital heart disease are fixed in size, requiring repeated open-heart surgeries during childhood to replace the valve with a larger version. But a surprising new design created at Boston Children's Hospital could allow children to keep the same prosthetic valve until adulthood, and could also benefit adults with heart valve defects. The new device is described in Science Translational Medicine, published online February 19.
Benchtop studies, computer simulations and extensive testing in large animal models demonstrate that the new prosthetic valve design works across a wide range of sizes, and that the valve retains its functionality when expanded via a minimally invasive balloon catheter procedure.
"We hope to bring this new device into clinical testing fairly rapidly," says Pedro J. del Nido, MD, Chairman of Cardiovascular Surgery at Boston Children's Hospital and senior author on the paper. "If our preclinical results hold up in human testing, this could transform the field."
Less is more: Two valve leaflets versus three
More than 330,000 children worldwide are born with a heart valve defect, and millions of others develop rheumatic heart disease requiring early valve replacement. Current prosthetic heart valves are fixed in diameter, so typically need to be replaced every few years; children receiving their first replacement before age 2 will need as many as five high-risk open-heart operations before reaching adulthood.
Commercially available prosthetic heart valves have three leaflets, tiny flaps that provide a one-way inlet or outlet for blood to keep it flowing in the right direction. The new design was inspired by human venous valves, located in the deep veins of the leg. Unlike our hearts' native outflow valves, our venous valves have just two leaflets, and a geometry that is optimized to maintain closure and one-way flow even when the veins expand in diameter to accommodate larger volumes of blood passing through.
-
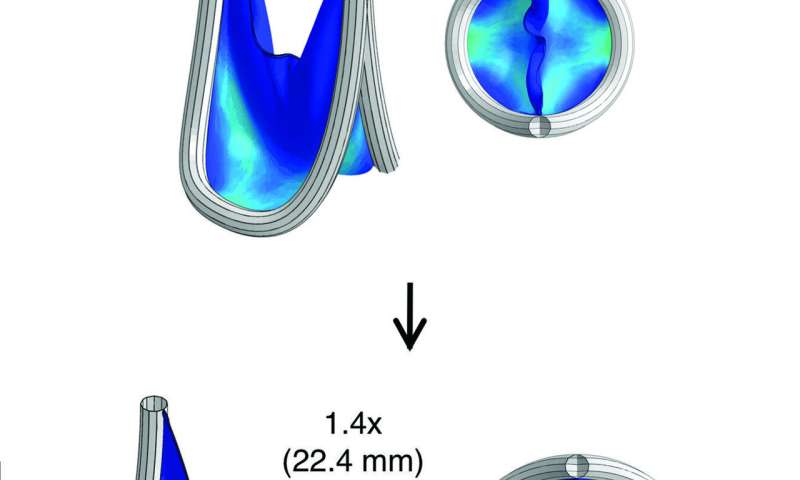
The scientists used computational modeling to predict how their valve replacement expanded to deal with the stress of blood flow. Credit: S.C. Hofferberth et al., Science Translational Medicine (2020) -
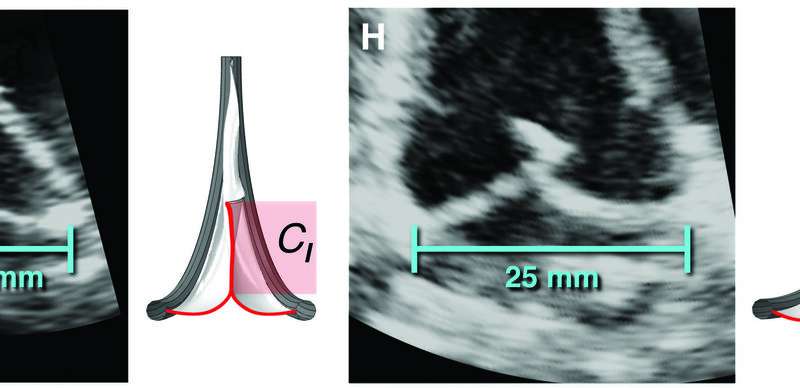
The valve replacement expanding to accommodate different lengths in implanted sheep. Credit: S.C. Hofferberth et al., Science Translational Medicine (2020) -
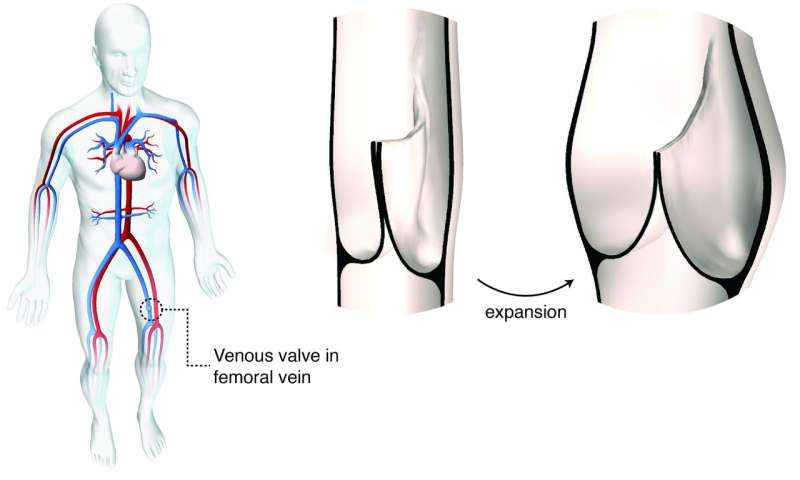
Artistic rendering of human venous valve geometry at rest and during expansion under volume load. Credit: Sophie C. Hofferberth, Lara Tomholt
"Veins carry approximately 70 percent of our blood volume," says Sophie C. Hofferberth, MD, a surgical resident at Brigham and Women's Hospital who led the research in del Nido's lab at Boston Children's. "The vein dimensions can change dramatically depending on body position, yet the valves must remain functional. We mimicked the geometric profile of the human venous valve to design a bileaflet valve of programmed dimensions that is adaptable to growth without loss of one-way flow control."
In multiple rounds of testing, in both benchtop and large animal models, valve prototypes with the biomimetic two-leaflet design were able to expand to accommodate growth and structural asymmetries within the heart. The valves remained fully functional across a wide range of dimensions, at a range of pressure and flow rates.
Because the valve is designed to expand without requiring the frame and leaflet to stretch or enlarge, it is compatible with a range of off-the-shelf materials, the researchers say. The study showed the device could be effectively expanded at multiple timepoints in a growing animal model, using a minimally invasive balloon catheter approach.
Potential for fewer blood clots
The researchers also observed that their "geometrically adaptable" design encourages a favorable blood flow profile through the valve, potentially reducing the risk for blood clot formation often seen with existing valve replacement devices. In the growing sheep model, there was no evidence of blood clot formation over 10 weeks of observation, even without the use of blood-thinning medication typically given to prosthetic valve recipients.
"A shortcoming of many existing devices is the presence of flow disruptions that lead to blood clot formation and early valve deterioration," says Hofferberth, who is first author on the paper. "Our design achieves a favorable flow profile that seems to facilitate effective valve washout and minimize flow stagnation, which is likely to be an important determinant of long-term device durability."
The research team believes their data support initiation of a clinical study within one to two years.
More information: S.C. Hofferberth el al., "A geometrically adaptable heart valve replacement," Science Translational Medicine (2020). stm.sciencemag.org/lookup/doi/ … scitranslmed.aay4006