Positive outcome for Hendra antibody trial

A process developed by University of Queensland researchers to produce larger quantities of the Hendra virus therapeutic antibody could be expanded to manufacture treatments for other potentially deadly viruses around the world.
Researchers at UQ's Australian Institute for Bioengineering and Nanotechnology (AIBN) used this process to produce material for Queensland Health's world-first human Hendra virus clinical trial.
Professor Trent Munro, Director of the National Biologics Facility (NBF) based at AIBN, said the ability to produce anti-bodies at larger scales, coupled with formal regulatory approval via clinical trials, will play an important role in mitigating the impact and spread of these diseases.
"Working with Queensland Health, Uniformed Services University of the Health Sciences (USU) and the Henry M. Jackson Foundation for the Advancement of Military Medicine (HJF), we obtained an antibody-producing cell line and then developed an optimized process to produce the larger amounts required for human clinical use," Professor Munro said.
"We're very fortunate to have a facility such as UQ's NBF as it offers a unique capability to manufacture these novel products—normally this would only be possible at great expense and long timelines within a commercial facility."
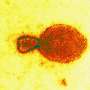
Hendra is a rare but deadly henipavirus that infects fruit bats, however it can be transmitted to horses, and then passed on to humans who have had close contact with an infected horse.
The therapeutic antibody, m102.4, developed by Professor Chris Broder and his team at the USU and the National Institutes of Health in the US, blocks the virus' entry to healthy human cells enabling the immune system to fight it off.
It was originally used on compassionate grounds, in a small number of people who had experienced a high risk exposure to the virus, however it was unclear if the use of the monoclonal antibody influenced the outcome.
Professor Munro said that there was potential to use the antibody against another henipavirus such as the lethal Nipah virus—listed by the World Health Organisation as a priority pathogen with epidemic potential.
"There are regular outbreaks of Nipah in Asia, so having a treatment option in these countries will be critical," he said.
"The ability to produce the m102.4 antibody at increasingly larger scales coupled with formal regulatory approval via clinical trials will play an important role in mitigating the impact and spread of these diseases.
"Enabling quick, technology-driven solutions to very serious disease outbreaks is an area where we need continued investment, or we risk not being able to respond appropriately in the future."
The results of the trial, which was funded by Queensland Health, the National Health and Medical Research Council and the National Hendra Virus Research Program, have been published in The Lancet Infectious Diseases journal.
More information: Elliott Geoffrey Playford et al. Safety, tolerability, pharmacokinetics, and immunogenicity of a human monoclonal antibody targeting the G glycoprotein of henipaviruses in healthy adults: a first-in-human, randomised, controlled, phase 1 study, The Lancet Infectious Diseases (2020). DOI: 10.1016/S1473-3099(19)30634-6