Lifestyle changes could delay memory problems in old age, depending on our genes
Researchers from King's College London have shown that how we respond to changes in nutrients at a molecular level plays an important role in the aging process, and this is directed by some key genetic mechanisms.
Published in Communications Biology, the study explored the molecular interactions that occur in our response to varying levels of nutrients, otherwise known as nutrient-sensing pathways, as we grow older.
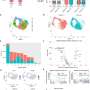
Using a combination of laboratory-based techniques and analysis of data on people's memory, diet and level of physical exercise, researchers identified a number of genes that are active in nutrient-sensing pathways and demonstrated these genes also show associations to performance on memory tasks in data from over 2000 individuals. The study identified that the genes ABTB1 and GRB10 were both influential in nutrient sensing pathways and showed association to memory.
Advances in medicine and healthcare over the past century have led to increased life expectancies. However, ageing is still accompanied by frailty and a decline in our thought processes. This level of decline varies enormously across individuals and an improved understanding of what influences these ageing mechanisms could help develop strategies to increase "healthspan", which is the period of time free from debilitating disease.
Previous studies have indicated that neural stem cells (NSCs) in the hippocampal part of the brain play an important role in the decline of our thought process and memory over time. NSCs are cells that keep dividing as long as they are alive and either make more NSCs or cells that have a specialist function in the brain. The maintenance of these NSCs is important in memory and is affected by environmental factors such as diet and exercise, potentially explaining some of the variation in how ageing affects different people.
Although the role of nutrient-sensing-pathways in ageing and the maintenance of stem cells in the brain has been investigated in animal models, no human studies have so far investigated their role in NSCs in the hippocampus.
The study aimed to explore whether nutrient-sensing pathways can provide the molecular basis for the association between lifestyle and ageing. These pathways have been implicated in stem cell maintenance, suggesting they could also be involved in the interaction between lifestyle, NSCs and cognition. Using a novel back-translation approach which uses laboratory-based experiments on NSCs to inform analysis of epidemiological data rather than vice versa the researchers showed that variations in ABTB1 are associated to performance on a standard memory task and that variation in the gene GRB10 is an important player in determining the association between Mediterranean diet and memory performance. The study also identified an interaction between exercise levels and the SIRT1 genotype which led to different performances on memory tasks.
Lead author, Chiara de Lucia, from the Institute of Psychiatry, Psychology & Neuroscience (IoPPN), King's College London said: 'Our study shows that nutrient-sensing pathways play an important role in memory and suggests that the ABTB1 and GRB10 genes are likely molecular links for the association between diet, the ageing of neural stem cells and our memory ability. Identifying these genes as the missing links between these three important variables could inform new approaches to help improve the ageing process through targeted changes in diet and exercise and ultimately in developing new drugs in the future.'
Senior author, Sandrine Thuret from the IoPPN said: 'Finding the means to prevent or slow down the processes that drive the decline of our thought processes during ageing is one of the great endeavours of the 21st century. To our knowledge this is one of the first studies looking at these relationships with human data and adopting this back-translation approach which uses lab-based experiments to inform research on large datasets, allowing for a more targeted approach.'
'Our findings suggest that changes in lifestyle may be able to delay a decline in memory and thinking but that the effectiveness of these approaches will depend on the genetic makeup of each person. For example, adherence to a diet such as the Mediterranean diet may be most beneficial for people with a specific GRB10 mutation while increased exercise may be a better approach for participants with specific SIRT1 variations. Future research should look to replicate these findings on a larger dataset which would allow for the testing of 3-way interactions between diet, exercise and memory to gain a more comprehensive understanding of how these relate to one another.'
Researchers analysed the molecular and genetic mechanisms by exposing human NSCs to serums from either young or old individuals and to chemicals whose effects mimic the ageing process. The genes identified from the in vitro analysis as important in nutrient-sensing-pathways were then associated with genetic data from over 2000 individuals from the TwinsUK cohort and data on performance on the Paired Associates Learning(PAL) task which assesses visual memory and learning, healthy eating, Mediterranean diet, calorie intake and physical activity.
More information: De Lucia, C. (2020) Lifestyle mediates the role of nutrient-sensing pathways in cognitive aging: cellular and epidemiological evidence, Communications Biology. DOI: 10.1038/s42003-020-0844-1