Inhibiting thrombin protects against dangerous infant digestive disease
Necrotizing enterocolitis (NEC), a rare inflammatory bowel disease, primarily affects premature infants and is a leading cause of death in the smallest and sickest of these patients. The exact cause remains unclear, and there is no effective treatment. No test can definitively diagnose the devastating condition early, so infants with suspected NEC are carefully monitored and administered supportive care, such as IV fluids and nutrition, and antibiotics to fight infection caused by bacteria invading the gut wall. Surgery must be done to excise damaged intestinal tissue if the condition worsens.
A new preclinical study by researchers at the University of South Florida Health (USF Health) Morsani College of Medicine and Johns Hopkins University School of Medicine offers promise of a specific treatment for NEC, one of the most challenging diseases confronting neonatologists and pediatric surgeons. The team found that inhibiting the inflammatory and blood-clotting molecule thrombin with targeted nanotherapy can protect against NEC-like injury in newborn mice.
Their findings were reported May 4 in the Proceedings of the National Academy of Sciences.
"Our data identified the inflammatory molecule thrombin, which plays a critical role in platelet-activated blood clotting, as a potential new therapeutic target for NEC," said coauthor Samuel Wickline, MD, professor of cardiovascular sciences at Morsani College of Medicine and director of the USF Health Heart Institute. "We showed that anti-thrombin nanoparticles can find, capture and inactivate all the active thrombin in the gut, thereby preventing or reducing the small blood vessel damage and clotting that accelerates NEC."
The PNAS paper's senior author is Akhil Maheshwari, MD, professor of pediatrics and director of neonatology at the Johns Hopkins University School of Medicine. Before joining Johns Hopkins Medicine (Baltimore) in 2018, Dr. Maheshwari's group at USF Health was the first to demonstrate that platelet activation is an early, critical event in causing NEC, and therapeutic measures to block these platelets might be a new way to prevent or reduce intestinal injury in NEC.
The nanotherapy platform created by Dr. Wickline and USF Health biomedical engineer Hua Pan, Ph.D., delivers high drug concentrations that specifically inhibit thrombin from forming blood clots on the intestinal blood vessel wall without suppressing the (clotting) activity needed to prevent bleeding elsewhere in the body. This localized treatment is particularly important for premature infants, Dr. Wickline said, because the underdeveloped blood vessels in their brains and other vital organs are still fragile and susceptible to rupture and bleeding.
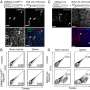
For this study the researchers used a model they created—infant mice, or pups, induced to develop digestive tract damage resembling human NEC, including the thrombocytopenia commonly experienced by premature infants with NEC. Thrombocytopenia is characterized by low counts of blood cell fragments known as platelets, or thrombocytes, which normally stop bleeding from a cut or wound by clumping together to plug breaks injured blood vessels.
The molecule thrombin plays a key role in the bowel inflammation driven by overactive platelets. While investigating role of platelet depletion in NEC-related thrombocytopenia, the USF-Johns Hopkins researchers were surprised to find that thrombin mediates platelet-activated blood clotting early in the pathology of NEC-like injury—before bacteria leaks from inside the gut to circulating blood or other organs. This clotting clogs small blood vessels and restricts blood flow to the inflamed bowel. Eventually, the lining of the damaged intestinal wall can begin to die off.
The investigative therapy essentially works "like a thrombin sponge" that is exponentially more potent than current agents used to inhibit clotting, Dr. Wickline explained. "It literally puts trillions of nanoparticles at that damaged (intestinal wall) site to sponge up all the overactive thrombin, which tones down the clotting and inflammation processes promoted by thrombin."
"We are so excited about finding this new way to attenuate intestinal injury in NEC," Dr. Maheshwari said.
The same approach has also been shown in preclinical studies to inhibit the growth of atherosclerotic plaques and certain kidney injuries without causing systemic bleeding problems. Dr. Wickline added. "The nanoparticles can be tailored to other inflammatory diseases highly dependent on thrombin for their progression."
The study authors conclude that their experimental targeted treatment for NEC merits further evaluation in clinical trials.
More information: Kopperuncholan Namachivayam et al, Targeted inhibition of thrombin attenuates murine neonatal necrotizing enterocolitis, Proceedings of the National Academy of Sciences (2020). DOI: 10.1073/pnas.1912357117