New biomaterial has potential to repair damaged bone with lower risk of inflammation

Scientists at RCSI University of Medicine and Health Sciences have developed a new biomaterial that has the potential to accelerate bone regeneration by promoting an immune response that encourages repair and lowers the risk of inflammation.
The study, conducted by researchers at RCSI Tissue Engineering Research Group (TERG) and AMBER, the SFI Research Centre for Advanced Materials and BioEngineering Research, is published in Acta Biomaterialia
The researchers have developed a technology that is a combination of nanoparticles and a collagen-based biomaterial called a scaffold, specifically designed by RCSI TERG that can be surgically implanted to aid bone tissue repair. The material allows for the delivery of a microRNA silencer, a molecule capable of influencing the way our cells function.
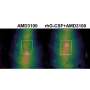
In laboratory conditions, researchers successfully demonstrated that damaged bone tissue is restored as the particular microRNA delivered by the biomaterial works to increase cells responsible for bone repair. The technology also assists in promoting a pro-repair immune system response, lowering the risk of inflammation and other complications.
"The results of our research are a promising step towards improving health outcomes for patients with fractures that fail to repair naturally or have degenerative bone diseases such as osteoporosis, although further pre-clinical and clinical trials are still required before the technology could be used to treat humans," said Dr. Caroline Curtin, Lecturer in Anatomy and Regenerative Medicine at RCSI.
"We are confident that this biomaterial system will have several potential applications beyond bone repair, as it can be tailored to deliver other therapeutic molecules that address degenerated or diseased tissue in the body. At RCSI Tissue Engineering Research Group, we are exploring these possibilities through the development of similar methods to repair articular joints like the knee and hip, and attempting to apply the microRNA delivery systems to inhibit breast cancer cell growth and other novel research," said Prof. Fergal O'Brien RCSI Director of Research and Innovation, Professor of Bioengineering and Regenerative Medicine and Deputy Director of the SFI AMBER Centre.
More information: Irene Mencía Castaño et al, Rapid bone repair with the recruitment of CD206+M2-like macrophages using non-viral scaffold-mediated miR-133a inhibition of host cells, Acta Biomaterialia (2020). DOI: 10.1016/j.actbio.2020.03.042