Desperately seeking a COVID-19 vaccine—the race is on
Seven months after the emergence of the worst global health crisis in a century the race is on to find the medical equivalent of the Holy Grail—a COVID-19 vaccine.
To date the novel coronavirus has killed about 590,000 people worldwide and infected close to 14 million in 196 countries since the outbreak first emerged in China late last year.
How to stop in its tracks a pandemic which has wreaked havoc on the world economy is now the pressing multi-billion-dollar issue for vaccine developers aware that every suggestion the goal may be in sight will be pored over in microscopic detail.
How many vaccines?
In its most recent assessment this month, the World Health Organization identified 21 "vaccine candidates"—almost half involving Chinese companies or institutes—following a series of global clinical trials.
Many are still in what is termed the first phase, which seeks to establish a product's security and dosage,
Some however have graduated to phase two, which examines how effective a medicine may be.
Two candidate vaccines have reached stage three, whereby developers monitor issues such as degree of potential toxicity on a much larger scale and test it in a wider range of conditions prior to eventual submission for approval.
The duo concerned are a European project being developed at Oxford University in tandem with AstraZeneca and a Chinese variant from biopharmaceutical company Sinovac in collaboration with Brazilian research institute Butantan.
Sinovac's project is among the most advanced—it will start the testing for its vaccine on volunteers in Brazil this month.
A Canadian clinical trial to involve CanSino Biologics was also earlier given the green light, and the company said last month that China's military had approved for use within its ranks the vaccine it helped to develop.
Sinopharm, which is working with the United Arab Emirates for trials, finished construction of a new production facility with a capacity of 100 million doses on July 1, Chinese authorities said on social media.
Beyond the tests already under way the WHO is monitoring a further 139 potential vaccines which are still at the pre-clinical evaluation stage, involving testing on animals.
Britain, Canada and the United States this week accused Moscow-linked hackers of targeting labs in their countries conducting coronavirus vaccine research.
Which techniques?
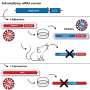
There's a variety of approaches and techniques across proven or experimental vaccines.
Some teams work on inactivated "classic" vaccines which use a virus germ that has been "killed" while others, known as attenuated or weakened varieties, involve using a live virus rendered less virulent.
Other types still include "sub-unit" vaccines, which contain a fragment of the pathogen that it is derived from to produce an appropriate immune response.
Then there are more innovative "viral vector" varieties which use live viruses to deliver DNA into human cells to trigger an immune response.
An example is to take a virus such as measles and, as the Pasteur Institute is doing, modify it with a coronavirus protein and deploy it against COVID-19.
Then there are new experimental developments involving gene-based DNA or RNA vaccines using fragments of modified genetic material.
"The more candidate vaccines there are, and above all the more types of candidate vaccines, the greater the chance of getting somewhere" in terms of beating the virus, says Daniel Floret, vice-president of France's Technical Vaccination Committee.
Results so far
For now, only partial results have been made public, some dubbed promising by the firms behind them.
Jean-François Delfraissy, chairman of France's scientific council which has been guiding the French authorities on the issue throughout the pandemic so far, said claims have to be closely probed as the commercial effect of a potential, but unconfirmed, breakthrough can be huge.
"There are considerable effects on the pharmaceutical industry from an announcement which one has to view with the utmost caution," the immunologist told AFP.
"A vaccine injection (tested on) 30 people which gets announced as a result—well, that's not a result," Delfraissy noted.
Floret agreed that many announcements are directed at least as much at the stock market as at the general public.
"It's never neutral. It is important they show us that they are speeding things up but shall all the same have to see the results and for the moment that's not happening."
Going fast?
All the while, with the pressure on to find a means of dealing with the pandemic, procedures have been ramped up worldwide like never before.
"Things are advancing at a rate of knots... and projects are going along perhaps a bit quicker than forecast," Christophe d'Enfert, from the Pasteur Institute, told AFP.
States and large foundations alike have been falling over themselves to push fundraising efforts.
And while the United States has been looking to go it alone, Europe and most other countries are looking at cooperation.
The Trump administration has set up "Operation Warp Speed" in a bid to deliver 300 million doses, essentially for US citizens, of a safe, effective vaccine for COVID-19 by January.
Firms "are trying to develop a vaccine and at the same time put in place an industrial process to produce it whereas normally you wait to see the results" of the first of those operations, notes Delfraissy.
"That's why they are calling on states for financing, saying: 'We are taking a risk starting off on (developing) a vaccine which maybe won't work and if we put in place the industrial (production) process we shall have to be partially covered by international financing."
Security problems
"To authorise any COVID-19 vaccine, EMA will need to have strong evidence from clinical trials on the safety, efficacy and the quality of this vaccine," warns the European Medicines Agency, an EU body.
Forcing the pace in clinical trials "can cause problems" regarding security, says Floret, who says it is "key to have proof that the vaccine is not liable to exacerbate the illness".
That, he recalls, did happen in some tests on monkeys "during vaccine development tests for MERS-CoV and SARS," other strains of coronavirus.
A similar problem arose in terms of some measles tests on humans in the 1960s which were withdrawn as well as some for bronchiolitis that were likewise abandoned, says Floret.
How long?
EMA warns that "vaccine development timelines are difficult to predict".
Based on previous development timelines, it said it may take until next year before a COVID-19 vaccine is ready for widespread use.
Even so, some firms are optimistic they can turn up trumps before the end of this year.
In Floret's view, "I am not that sure it is very realistic (to say) a vaccine will be there by the autumn—we have to temper such enthusiasm."
He believes the first quarter of next year is likelier and "if we get there by then we shall already have done extremely well," given the usual development timeline is several years.
How about never?
The world is now awaiting a vaccine as it were a kind of messiah as the only infallible means of defeating the pandemic.
But what if one never arrives?
Delfraissy said: "We have never perfected a vaccine against a coronavirus, even if we have never completely given ourselves the means to do so.
"There is a degree of uncertainty over our capacity to perfect a vaccine against coronavirus,—but all the elements to get there are in place," he stressed.
"Everything is possible, the maximum means and different techniques envisaged are out there and it would be astonishing if we didn't get there," Floret said.
Yet even if the researchers arrive at the finish line a large question will still remain—will people accept being vaccinated in a world where there is growing mistrust of vaccination?
Perhaps not.
"As repeated measles outbreaks demonstrate, we haven't done a great job addressing people's concerns about vaccines. And if we don't learn from our failed response to them, a coronavirus vaccine program will be doomed," Phoebe Danziger, a paediatrician at the University of Michigan, told the New York Times.
© 2020 AFP