Studying brain plasticity in old age
Neuronal critical periods are limited phases of life during which the plasticity of neural connections is at its maximum and the development of the brain is adapted to the environment. Researchers at the Scuola Normale Superiore in Pisa and the Leibniz Institute on Aging (FLI) in Jena have discovered the role of a small microRNA (miR-29) in these learning-dependent phases of plasticity. A premature increase in miR-29 concentration in young mice blocks cortical plasticity, whereas blocking miR-29 in adult animals induces plasticity typical for younger sensitive phases; an indication that miR-29 is an age-dependent regulator of developmental plasticity.
Postnatal development of the brain after birth is characterized by temporary, function-specific windows of high plasticity (critical or sensitive periods). During these phases, certain areas of the brain further develop through several maturation and differentiation processes, where neuronal connections quickly create and increase the brain's plasticity. Natural language acquisition in infants is the best-known example of such a sensitive phase.
Neuronal plasticity gives our brain the ability to adapt to new demands throughout our lives. However, this is often limited in the adult brain, so that learning processes are more laborious. To identify cellular and molecular mechanisms that open and close these sensitive phases in relation to aging, researchers of the Scuola Normale Superiore (SNS) in Pisa, Italy, in cooperation with the Leibniz Institute on Aging – Fritz Lipmann Institute (FLI) in Jena, Germany, and other partners, studied the plasticity of the visual cortex in mice.
Plasticity of the visual cortex
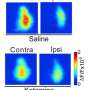
"Neuronal nets in the visual cortex adapt to visual stimuli to the maximum extent in sensitive developmental phases. This allows us to identify important regulators of brain plasticity," say the senior authors of the study published in EMBO Reports, Prof. Alessandro Cellerino, SNS and associated group leader at FLI in Jena and Prof. Tommaso Pizzorusso, University of Florence and Institute of Neuroscience, NRC, in Pisa. One paradigm posits that plasticity of ocular dominance passively decays with age; however, it is becoming increasingly clear that plasticity levels are set by the coordinated action of age- and experience-dependent molecular processes that either actively promote or suppress circuit plasticity.
"Visual cortical circuits, which are part of the visual system and enable vision, exhibit strong plasticity during early life and are later stabilized by molecular brakes that limit excessive adaptation of links beyond a critical period," explains Prof. Cellerino. However, the underlying mechanisms that coordinate the expression of these factors during the transition from development to adulthood remain unknown.
MiR-29 - age dependent control of plasticity in the visual cortex
To identify factors that regulate postnatal development of the visual cortex, the research team analyzed miRNA/RNA data sets from the developing visual cortex of mice. They compared them at different time points: P10, day 10 after birth and immediately before eye opening and the onset of the sensitive phase, and P28, when the mouse cortex has reached functional maturity.
Their results showed the microRNA family miR-29 is an age-dependent regulator of developmental plasticity in the visual cortex. "With a 30-fold increase, miR-29a was the most highly upregulated miRNA during the sensitive phase," explained Prof. Cellerino from FLI/SNS. The regulation of the miR-29 family is strikingly conserved in fish, mice and humans. In addition, more than half of the targets regulated by miR-29 are down-regulated with age, including key regulators of brain plasticity. This indicates that miR-29a is an important regulator of downstream developmental processes.
Remodeling plasticity
Further analysis showed that a premature increase in miR-29a concentrations in young mice blocked juvenile eye dominance plasticity and caused an early appearance of perineural nets (PPNs). PPNs are specialized structures in the central nervous system that are responsible for synaptic stabilization in the adult brain. In the developing as well as in the adult brain they play a crucial role to break plasticity and maintain existing connections between nerve cells.
In addition, the researchers were able to show that blocking miR-29a in adult animals reversed the developmental downregulation of miR-29a targets and induced a form of ocular plasticity with a typical physiological and molecular signature of plasticity in sensitive phases.
In summary, the data now published in the journal EMBO Reports indicate that miR-29a is an important regulator of plasticity breaks that promotes the age-related stabilization of visual cortical connections. The observation that miR29a is a remodeler of mature neural networks opens new and promising therapeutic perspectives for miR-29a and other miR-29 family members for promotion of brain plasticity during aging and regeneration of brain damages.
More information: Debora Napoli et al. MiR‐29 coordinates age‐dependent plasticity brakes in the adult visual cortex, EMBO reports (2020). DOI: 10.15252/embr.202050431