Researchers develop 3-D dressing to heal wounds
Researchers at ITMO University and the University of Toronto have developed a material based on gelatin and nanocrystalline cellulose that can fight off antibiotic-resistant bacteria and speed up the wound healing process. In the future, the material could be used in the production of gel-based wound dressing. An article on the results of the research was published in Chemistry of Materials.
Each use of antibiotics results in the surviving bacteria developing a resistance to antibiotics, a trait which they pass to their offspring. This results in super-bacteria that are very difficult to treat. These may be a source of serious health complications which is why researchers around the world are looking for new ways to fight infections without using antibiotics. This is especially relevant when it comes to wound treatment and wound healing, as a poorly treated wound increases the likelihood of severe infection.
"A wound dressing must possess antibacterial qualities in order to, at the very least, not cause an infection. There is now a global trend of reducing the use of conventional antibacterial products due to bacteria developing antibiotic resistance. We have suggested an alternative way of dealing with this issue," says Dr. Elena Koshel, an associate professor at ITMO University's ChemBio Cluster.
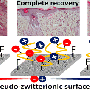
Working within the framework of a joint grant project, researchers at ITMO University and the University of Toronto have proposed using biomaterials to create dressings that would aid the healing process. They have developed a hydrogel that facilitates the healing process by providing favorable conditions for cell growth; preventing bacteria reproduction; and indicating when the dressing must be changed.
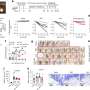
The basis of the material is modified nanocrystalline cellulose and gelatin. The components are linked using a special method of chemical grafting which ensures a strong bond between them. Using cellulose and gelatin makes it possible to create a layer that protects the wound while providing favorable conditions for natural healing. In addition, the gel's damp and soft texture ensures that changing the dressing would not cause further damage to the wound.
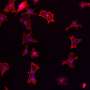
"Because cellulose is able to adsorb iron (III) ions which provide nutrition to bacteria, the material prevents the spread of infection. Furthermore, by modifying it with carbon nanodots, we have made it optically active—exposure to ultraviolet light causes it to emit a glow in the blue band of the spectrum. If the bandage has yet to exhaust its ability to adsorb ions, it will produce an optical response; if it is completely covered in ions, no response will be produced," explains Dr. Elena Krivoshapkina, an associate professor at ITMO University's ChemBio Cluster.
More information: Mahshid Chekini et al. Nanocolloidal Hydrogel with Sensing and Antibacterial Activities Governed by Iron Ion Sequestration, Chemistry of Materials (2020). DOI: 10.1021/acs.chemmater.0c03362