Molecular interactions regulating trans-synaptic signalling and synapse formation

Scientists at Korea's Daegu Gyeongbuk Institute of Science and Technology (DGIST) and colleagues have uncovered some of the complex molecular mechanisms involved in the formation of the brain's neural circuits. Their findings were published in The Journal of Neuroscience and could be relevant for developing treatments for diseases, such as autism spectrum disorders and schizophrenia.
Synaptogenesis is a complex molecular process that promotes the growth and development of nerve fiber ends so they can recognize and communicate with their appropriate nerve fiber partner via molecules transmitted across specialized junctions, called synapses.
"A comprehensive understanding of synaptogenesis is essential for designing therapeutic approaches against many devastating brain disorders," says DGIST neuroscientist, Professor Jaewon Ko. "So it's really crucial to develop fine-tuned molecular manipulations that can target key synapse components in order to understand their roles."
Professor Ko and his team of scientists specifically looked at two key 'synaptic adhesion molecules' involved in synaptogenesis. Neurexins and leukocyte common antigen-related protein tyrosine phosphatases (LAR-RPTPs) are transmembrane proteins that are located on the 'presynaptic' sending side of a developing nerve junction. They are known to be involved in the formation and maintenance of synapses. But it has been unclear whether they cooperate with each other and how they interact with other synaptic molecules in regulating synapse organization.
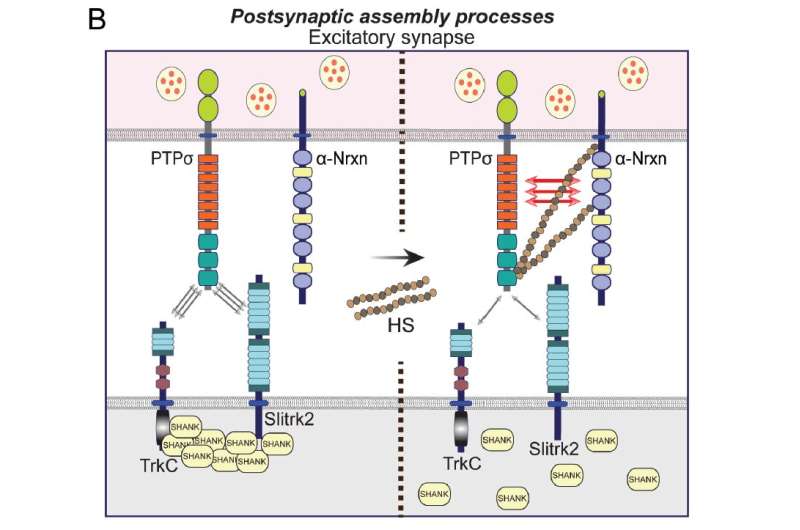
To address these questions, the scientists conducted a series of extensive experiments in rodent nerve cell cultures and then in fruit flies in which Drosophila orthologs of neurexins and LAR-RPTPs were deleted. They observed that two members of LAR-RPTPs (termed PTPσ and PTPδ) are required for neurexins to promote presynaptic differentiation. At the sending end of the developing synapse, neurexins bind to either of the two LAR-RPTPs through a distinct set of molecules depending on whether the synapse will be excitatory or inhibitory; in other words if it will send signals to activate or to turn off its receiving nerve end.
The scientists also found that PTPσ and neurexin directly interact through specific glycans, called heparan sulfates, to direct the formation of the receiving nerve end at excitatory synapses.
"We believe that our findings have significance in terms of proposing a novel molecular model underlying synapse organization, and possibly have implications for understanding neural circuit architecture and brain functions," says Professor Ko.
The team is now investigating downstream mechanisms underlying interactions of neurexins with LAR-RPTPs in vertebrate neurons, and is working to pinpoint a set of intracellular proteins involved in trans-synaptic signaling in presynaptic neurons. Further studies are essential before these findings can be translated into clinical studies.
More information: Kyung Ah Han et al, LAR-RPTPs Directly Interact with Neurexins to Coordinate Bidirectional Assembly of Molecular Machineries, The Journal of Neuroscience (2020). DOI: 10.1523/JNEUROSCI.1091-20.2020