Mexico approves emergency use of Pfizer coronavirus vaccine

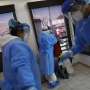
The Mexican government's medical safety commission approved the emergency use of the Pfizer-BioNTech coronavirus vaccine Friday, making Mexico the fourth country to do so.
Assistant Health Secretary Hugo López-Gatell said Mexico's approval came after Britain, Canada and Bahrain. The United States announced its own approval of the vaccine shortly after Mexico.
Mexico is set to receive 250,000 doses of the vaccine, enough for 125,000 people, because each person requires two shots. López-Gatell has said that front-line health workers will get the shots first.
Vaccinations are expected to begin as soon as next week. López-Gatell said the approval "is of course a reason for hope," though the initial rounds of shots are not nearly enough for Mexico's estimated 1.6 million health care workers.
López-Gatell downplayed reports of allergic reactions among some vaccine recipients in Britain, noting that "the immense, immense, immense majority of people in Mexico and the world will be able to take this vaccine."
Mexico posted a record increase of 12,253 coronavirus cases Friday, for a total of 1,229,379 infections during the pandemic.
Officials also reported 693 more deaths related to COVID-19, bringing the country's total to 113,019. However, even official estimates place the real death toll at closer to 150,000, due to Mexico's extremely low level of testing.
-

Workers load an order of red snapper for a client onto a cart at the La Nueva Viga seafood market, part of the Central de Abastos, the capital's main market amid the COVID-19 pandemic, in Mexico City, Wednesday, Dec. 9, 2020. With numbers of reported COVID-19 infections on the rise, shoppers and sellers worry how increased restrictions will affect their businesses during this year's holiday season. (AP Photo/Rebecca Blackwell) -

Workers wearing protective suits walk to take up positions to dispense antibacterial gel to passing shoppers and workers, inside the Central de Abastos, the capital's main market, in Mexico City, Tuesday, Dec. 8, 2020.(AP Photo/Rebecca Blackwell) -

A worker pulls a cart with an order of red snapper for a client at the La Nueva Viga seafood market, part of the Central de Abastos, the capital's main market, in Mexico City, Wednesday, Dec. 9, 2020, amid the new coronavirus pandemic. With numbers of reported COVID-19 infections on the rise, shoppers and sellers worry how increased restrictions will affect their businesses during this year's holiday season. (AP Photo/Rebecca Blackwell) -
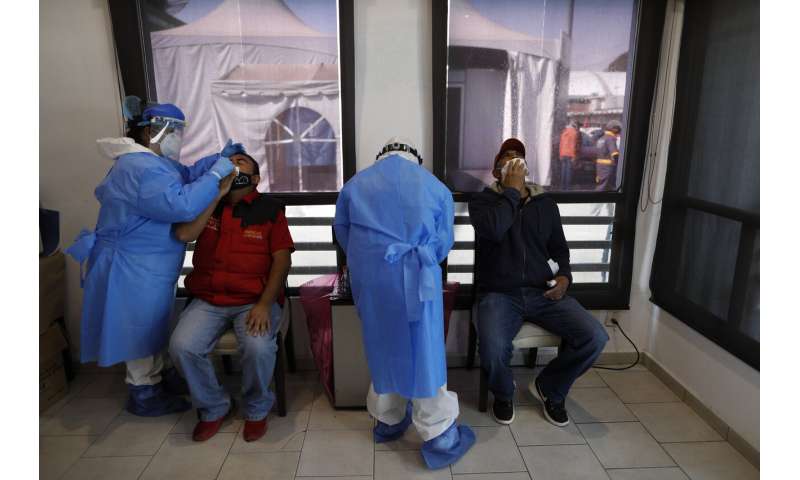
Medical staff collect nasal swabs for rapid and PCR tests inside a COVID-19 health post that has been set up in the Central de Abastos, the capital's main market, in Mexico City, Tuesday, Dec. 8, 2020.(AP Photo/Rebecca Blackwell) -

En elderly woman with her mask pulled down begs for change beside a worker offering antibacterial hand gel to passing shoppers and workers to curb the spread of the new coronavirus, inside the Central de Abastos, the capital's main market, in Mexico City, Tuesday, Dec. 8, 2020.(AP Photo/Rebecca Blackwell)
Mexico City has become the central focus of increasing cases in the country. The capital's hospitals were 78% full Friday, a level that worried officials.
Mayor Claudia Sheinbaum issued an urgent appeal for residents of the capital to stay home, saying the city of almost 9 million was in a "coronavirus emergency."
López-Gatell called it an "extremely urgent" situation.
© 2020 The Associated Press. All rights reserved. This material may not be published, broadcast, rewritten or redistributed without permission.