How fast could SARS-CoV-2 be detected?
In the past 20 years, humans have suffered several serious epidemics from emerging viruses, such as SARS, swine flu, Ebola, MERS and (most recently) SARS-CoV-2. During each epidemic, an accurate, rapid, and accessible molecular diagnostic test is highly essential for the control and prevention of viral diseases. In particular, coronavirus disease 2019 (COVID-19) is spreading rapidly in most countries, resulting in a severe global pandemic, which has a profound impact on the world economy and people's normal life. Accurate and rapid diagnosis of COVID-19 has been the most crucial measure for controlling the ongoing pandemic. In general, reverse transcriptase quantitative polymerase chain reaction (RT-qPCR) is the primary method for detection of nucleic acid-based genetic sequences. Because of the labor-intensive sample preparation, which must be conducted in a biological laboratory by professionals using specialized instruments, the typical turnaround time of the RT-qPCR method is longer than 24 hours. In addition, the complex RT-qPCR process might also reduce clinical sensitivity, resulting in false negative results.
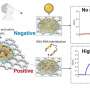
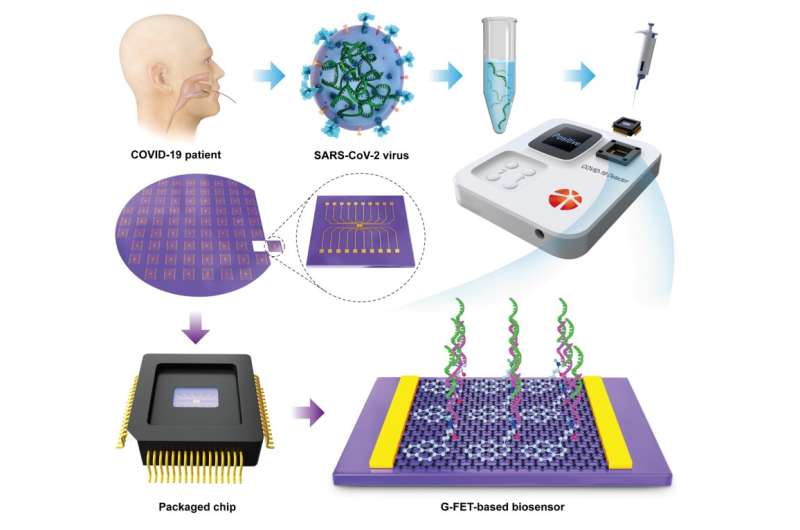
Recently, Guo and coworkers from Peking University developed an unprecedented accurate, rapid, and portable electrical detector based on the use of graphene field-effect transistors (G-FETs) for detection of RNA from COVID-19 patients. As shown in Fig. 1, the detection system mainly consists of two parts: a plug-and-play packaged biosensor chip and a home-developed electrical measurement machine. The unique feature of this method is that the extent of hybridization between the ss-DNA probe and viral RNA can be directly converted to the current change of graphene channels without repetition of the PCR process, thus affording an ultra-low limit of detection (LOD) of ~0.1 fg/mL for the detection of the RNA-dependent RNA polymerase (RdRp) gene target of SARS-CoV-2. Furthermore, this method was validated using clinical samples collected from many patients with COVID-19 infection and healthy individuals as well, and the testing results were in full agreement with those of PCR-based optical methods. The entire process, precluding the extraction of detection targets from oropharyngeal swabs, requires approximately 10 min. Because it does not involve time-consuming PCR step nor expensive instruments, this detection system enables massive point-of-care testing of COVID-19, outside of specialized diagnostic laboratories, with the advantage of high accuracy, sensitivity and low cost.
Notably, false negative results are inevitable in the course of nucleic acid testing; thus, the use of immunodetection as an auxiliary technique is important in the diagnosis of COVID-19 patients, especially those with suspected diseases. By replacing the ss-DNA probe with a SARS-CoV-2 antigen protein, this detection system can also detect SARS-CoV-2 IgM and IgG antibodies with an ultra-low LOD of ~1 fg/mL. Immunoassays of serum specimens of COVID-19 patients and healthy subjects matched excellently with those of PCR-based optical methods.
This detecting system exhibits obvious advantages of high sensitivity, rapid speed (~10 min for RNA analysis and ~5 min for immunoassay), and bifunction (both RNA analysis and immunoassay). These advantages enable high-throughput point-of-care testing, which may facilitate management of the current severe public health crisis. Furthermore, this detection system offers a universal methodology that is ready for immediate application in rapid detection of novel viruses in future.
More information: Guojun Ke et al, An accurate, high-speed, portable bifunctional electrical detector for COVID-19 SCIENCE CHINA Materials, DOI: 10.1007/s40843-020-1577-yv