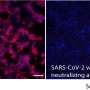
New insight into antibody-induced protective immunity to COVID-19
While PCR testing has been used widely for COVID-19 diagnosis, it only provides information on who is currently infected. Antibody testing can tell who has been previously exposed to SARS-CoV-2, the virus that causes COVID-19, a metric that is essential for tracking spread across a population. It may also, as a study recently published in the journal Nature Communications shows, hold the key to understanding the immune response to the virus.
Led by Galit Alter, Ph.D., Core Member of the Ragon Institute of MGH, MIT and Harvard, this study found that while antibodies against SARS-CoV-2 may be a good way to measure exposure to the virus, their presence alone wasn't enough to determine if a person had long-lasting protection. Instead, antibody effector functions associated with long-lasting protection, like virus neutralization and T cell responses, were only seen if the immune response included high levels of antibodies against a part of the virus called the receptor binding domain.
"Essentially," says Alter, who is also a professor of Medicine at Harvard Medical School (HMS), "this study indicates that it's not simply the presence or absence of antibodies that matter; rather, the amount and type of antibodies may play a defining role in the development of a protective immune response."
This data came from a cohort of adults who had mild or asymptomatic cases of COVID-19 (unusual among the studies that started in the early days of the pandemic), the result of a collaboration with SpaceX, an aerospace manufacturer and space transportation services company that was seeking data-driven ways to protect its essential workforce.
The project began in April of 2020 and, along with Alter and her group, included Anil Menon, MD, Medical Director at SpaceX; Doug Lauffenberg, Ford Professor of Biological Engineering, Chemical Engineering, and Biology at MIT; Elon Musk, CEO of SpaceX; and Eric Nilles, MD, Director of the Program on Infectious Diseases and Epidemics at the Harvard Humanitarian Initiative.
"This collaboration with SpaceX that has pulled together immunologists, epidemiologists, molecular and computational biologists and infectious disease modelers is a fantastic example of teamwork across institutions and disciplines, and I'm delighted to see this first publication from these efforts," says Nilles, who is also a professor at HMS and an attending physician at the Brigham and Women's Hospital.
Though the original goal of the study was to measure antibody levels over time, when reports of reinfection began surfacing, Alter's team realized their samples may hold much more valuable information than they originally thought.
"In early spring, we weren't sure if asymptomatic infection could drive long-lived antibodies," says Alter, "nor whether they possessed the capability to neutralize or kill the virus."
The team did know, however, that 120 of their study participants had experienced mild or asymptomatic COVID-19 which had resulted in the development of COVID-19 antibodies. By using sophisticated techniques to delve into these antibody responses, they discovered that individuals who had developed a larger number of antibodies, associated with stronger symptoms in case of mild COVID-19, had also developed immune functions associated with natural immune protection.
"Once you hit a certain threshold of these antibodies, it's like a switch turns on and we can observe antibody effector functions," says Yannic Bartsch, Ph.D., a Ragon postdoctoral fellow and first author on the study. "These functions were not observed in individuals with lower antibody binding titers, and the level of protection from reinfections is uncertain in these individuals."
Though more work remains to be done, this is an important first step in understanding natural immunity to SARS-CoV-2. With limited availability of COVID-19 vaccines, understanding asymptomatic infection, natural immunity, and who, exactly, is at risk for reinfection, is vital to an effective, successful global vaccine rollout.
More information: Nature Communications (2021). DOI: 10.1038/s41467-021-21336-8 , www.nature.com/articles/s41467-021-21336-8