Georgia begins vaccine rollout with AstraZeneca jab
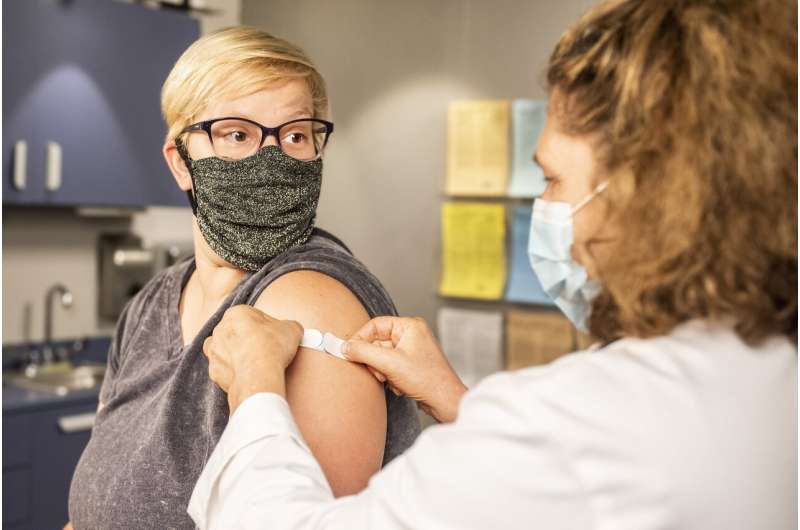
Georgia on Monday began a national coronavirus vaccine drive by inoculating medical workers with the AstraZeneca jab, rejecting concerns over side effects that have led several EU countries to halt its use.
The low-cost shot is crucial to rollouts in poorer parts of the world but several countries including the Netherlands, Denmark and Norway have paused inoculations with AstraZeneca over blood clot fears.
"We received some 43,000 doses of AstraZeneca through Covax and began today vaccinating medical staff," deputy director of the national centre for decease control Paata Imnadze told AFP, referring to the UN's mechanism for vaccine distributions.
"We are confident that AstraZeneca is safe. The World Health Organisation (WHO) and the European Medicines Agency (EMA) have said there is no evidence linking between the vaccine and clotting," he added.
Imnadze said Georgia expects to receive batches of the Pfizer/BioNtech vaccine in the near future and plans to inoculate 60 percent of the country's population by the end of the year.
The Black Sea nation of some 3.7 million people has so far reported 268,565 coronavirus cases and 3,650 deaths.
The WHO said last week there was no reason to stop using AstraZeneca, which is one of only three COVID-19 vaccines with the organisation's approval.
The EU's drug regulator, EMA, has said European countries could continue using the vaccine even though there were 30 cases of so-called thromboembolic events among five million people who had received the jab in Europe.
© 2021 AFP