Anti-tumor agent from the intestine
The microbiome is the vast collection of bacteria in the human gut. It is estimated that each person carries around 100 trillion bacterial cells in their digestive tract, belonging to several thousand species. These are believed to be involved in the development of chronic inflammatory intestinal diseases, to trigger diabetes, to be responsible for obesity, even neurological diseases such as multiple sclerosis and Parkinson's could have their causes here—not to mention depressions and autistic disorders.
The microbiome has been the focus of research for 20 years—ever since a new technique made it possible to analyze these bacteria quickly and precisely: high-throughput sequencing. Since then, there has been an increasing body of findings that the microbiome, which is sometimes also referred to as the second human genome, is not only of central importance for digestion, but also influences, if not controls, at least a large number of body functions. The immune system is mentioned particularly frequently.
The microbiome influences the immune system
Scientists at the Universities of Würzburg and Marburg have now succeeded for the first time in experimentally demonstrating that bacterial metabolites are able to increase the cytotoxic activity of certain immune cells and thus positively influence the efficiency of tumor therapies. Ideally, the composition of the bacterial species in the microbiome could be used to control its influence on the success of the therapy.
The research team published the results of its study in the journal Nature Communications. Dr. Maik Luu, postdoc in the laboratory of Professor Michael Hudecek at the Medical Clinic and Polyclinic II of the University Hospital of Würzburg, was responsible for the finding. Another participant was Professor Alexander Visekruna from the Institute of Medical Microbiology and Hygiene at the Philipps University in Marburg, where Luu did research before moving to Würzburg.
Fatty acids increase the activity of killer cells
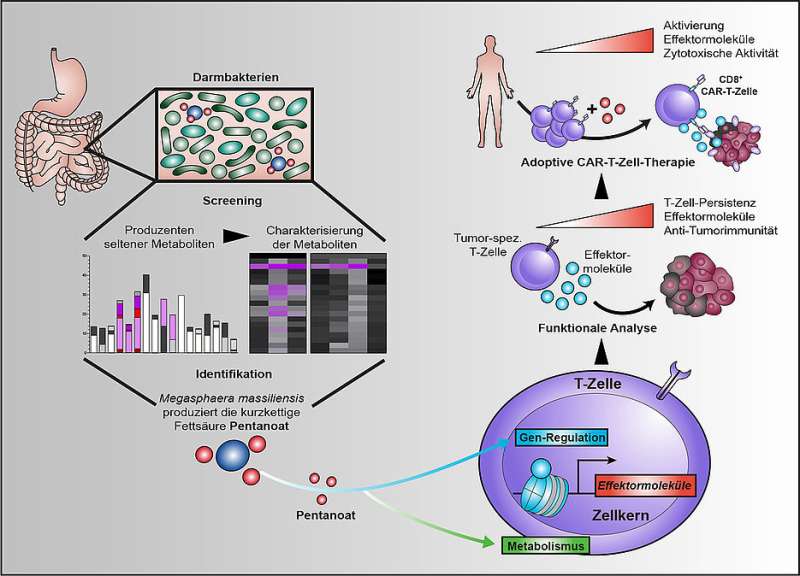
"We were able to show that the short-chain fatty acids butyrate and, in particular, pentanoate are able to increase the cytotoxic activity of CD8 T cells," Maik Luu describes the central result of the now published study. CD8 T cells are sometimes also called killer cells. As part of the immune system, it is their task to specifically kill cells that are harmful to the organism.
Short-chain fatty acids, in turn, belong to the most dominant class of metabolites of the gut microbiome. On the one hand, they can boost the metabolism of T cells by inducing central regulators of energy metabolism. On the other hand, they can inhibit specific enzymes that regulate the accessibility to the genetic material and thus the gene expression in the T cells. In doing so, they induce epigenetic changes.
Solid tumor models are combated more effectively
"When short-chain fatty acids reprogram CD8 T cells, one of the results is increased production of pro-inflammatory and cytotoxic molecules," Luu explains. In the experiment, treatment with the fatty acid pentanoate increased the ability of tumor-specific T cells to fight solid tumor models. "We were able to observe the same effect when fighting tumor cells with so-called CAR-T cells," says the scientist.
CAR-T cells are written out as "chimeric antigen receptor T cells". While normal T cells are largely "blind" to tumor cells, CAR T cells are able to recognize specific target antigens on the tumor surface and destroy the cancer cells thanks to a genetic modification. Michael Hudecek is one of the leading experts in the field of CAR-T cell research.
Targeted control via the composition of the microbiome
"The results are an example of how metabolites of intestinal bacteria can change the metabolism and gene regulation of our cells and thus positively influence the efficiency of tumor therapies," says Maik Luu. In particular, the use of CAR-T cells against solid tumors could benefit from this.
In these cases, therapy with genetically modified cells has so far been much less effective than the treatment of hematological tumors such as leukemia. This could change if the CAR-T cells were treated with pentanoate or other short-chain fatty acids before being used in patients, the scientists hope.
This effect might specifically be exploited via the composition of the bacterial intestinal colonization—especially since Luu and the others involved in the study were also able to identify the essential pentanoate producer of the intestinal flora: the bacterium Megasphaera massiliensis.
A long way to clinical applications
However, there is still a long way to go before the new findings will lead to new therapies for cancer patients. In a next step, the research team will initially expand the spectrum of tumor diseases investigated and, in addition to other solid tumors, also look at hematological tumor diseases such as multiple myeloma. In addition, it wants to investigate the functioning of short-chain fatty acids more intensively in order to identify starting points for targeted genetic modifications.
More information: Maik Luu et al, Microbial short-chain fatty acids modulate CD8+ T cell responses and improve adoptive immunotherapy for cancer, Nature Communications (2021). DOI: 10.1038/s41467-021-24331-1