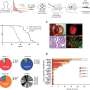
Pre-treatment levels of ctDNA may be useful in future clinical trials to predict long-term survival

Pre-treatment circulating tumor (ct)DNA levels more accurately predicted long-term survival than classical survival surrogates in the NADIM study and might be useful for the design of new clinical trials, according to research presented today at the IASLC 2021 World Conference on Lung Cancer. The study was presented in today's OA20: Exploring Treatment Modalities and Tools in Locally Advanced NSCLC.
There are currently no predictive biomarkers for long-term survival after neoadjuvant chemoimmunotherapy. However, the identification of patients with non-small lung cancer (NSCLC) who obtain long-term benefit from chemoimmunotherapy is essential to optimize therapies, said Dr. Atocha Romero, of the Hospital Universitario Puerta de Hierro, Majadahonda, Spain.
Using samples from NADIM clinical trial, in which patients with resectable stage IIIA NSCLC were treated with neoadjuvant chemoimmunotherapy with nivolumab, Dr. Romero and her co-researchers evaluated the capacity of ctDNA levels before treatment initiation to predict overall survival and progression-free survival. Dr. Romero compared its predictive value with classical survival surrogates as the pathologic response and clinical response assessed according to RECIST criteria v.1.1.
The ctDNA was analyzed by next-generation sequencing, using the Oncomine Pan-Cancer Cell-Free Assay. For each positive plasma sample, the sum of the mutant allele frequency (MAF) for all detected mutations was calculated. According to Dr. Romero, MAF (mutant allele fraction), VAF (variant allele fraction) or AF (allele frequency or allele fraction), refers to the amount of a given mutation detected in blood (in this case). It can be defined as the number of times a mutated base is read (by NGS), divided by the total number of times any base is observed at a given position in the genome. There is no standard procedure to calculate how much ctDNA a person has in blood, he said.
According to Dr. Romero, neither pathologic response nor clinical responses based on RECIST criteria were predictive for overall survival or progression-free survival. However, when excluding patients who died from COVID-19 (N = 2), pathologic complete response (but not major pathologic response) identified patients with improved PFS and OS. RECIST (Response Evaluation Criteria in Solid Tumours) provides a simple and pragmatic methodology to evaluate the activity and efficacy of new cancer therapeutics in solid tumors, using validated and consistent criteria to assess changes in tumor burden.
"On the contrary, in the multivariate analysis, patients with low ctDNA levels (< 1% MAF), in the baseline sample, had significantly improved progression-free survival and overall survival than patients in whom the opposite situation occurred," she said. (Adjusted HR: 0.22; 95%CI: 0.06-0.75; P = 0.016 and adjusted HR: 0.04; 95%CI: 0.00-0.45; P = 0.008 for PFS and OS, respectively).
The adjusted C-statistic (c) to predict PFS for ctDNA was 0.68 (95% CI: 0.51-0.84), which was superior to that of RECIST criteria (c = 0.61; 95% CI: 0.45-0.78) and similar to that of pathologic response (c = 0.68; 95% CI: 0.52-0.84). Similarly, baseline ctDNA levels predicted OS (c = 0.85; 95% CI: 0.72-0.99) better than RECIST criteria (c = 0.68; 95% CI: 0.44-0.93).
"Pre-treatment ctDNA levels more accurately predicted long-term survival than radiologic assessments in NADIM study and might be useful for the design of new clinical trials," Dr. Romero said.
More information: M. Provencio et al, OA13.05 NADIM Study: Updated Clinical Research and Outcomes, Journal of Thoracic Oncology (2019). DOI: 10.1016/j.jtho.2019.08.480