October 15, 2021 feature
A new way to prevent COVID-related clots and a biomarker that spots who's at highest risk
No aspect of SARS-CoV-2 infection has been more startling—or problematic—than the elevated risk for blood clotting, a concern that throughout the pandemic has been associated with severe COVID-19, frequently characterized by clotting events that have led to stroke, heart attack and organ damage.
But a team of medical scientists from a dozen institutions throughout the United States has made two key treatment-altering findings: Patients with moderate disease can also develop life-threatening clots, and need to be identified early and treated. The same team of medical investigators has pinpointed the most effective way to address the clotting problem, a finding that may have ramifications worldwide in the treatment of COVID-19 and its impact on the blood.
Dr. Alex Spyropoulos, a professor at the Feinstein Institutes for Medical Research in Manhasset New York, found that moderately ill patients hospitalized after a COVID diagnosis and who had elevated blood levels of a protein known as d-dimer, were at especially high risk for dangerous clots. Spyropoulos, principal investigator of the study discovered along with his team, that treating these patients with a high-dose of the blood thinner called low-molecular weight heparin (LMWH) significantly reduced the potential for clot formation and death.
The medication is readily available worldwide and is a cost-effective way to prevent clots and spare lives, the research found.
The results are expected to be practice-changing, affecting the way clinicians identify and treat hospitalized COVID patients—potentially reversing the course of the disease and increasing survival.
Reporting in JAMA Internal Medicine, Spyropoulos and colleagues sound a powerful note of caution–and hope–from the clinical study titled the HEP-COVID trial: Just because patients are hospitalized with moderate COVID symptoms doesn't mean they're at lower risk of life-threatening COVID-related blood clots.
"The HEP-COVID trial was able to identify an exquisite biomarker—very elevated d-dimer—that not only predicted a high risk COVID-19 inpatient population, but those whose risk was ameliorated by early use of therapeutic heparin anticoagulation for thromboprophylaxis," Spyropoulos told Medical Xpress, referring to clot prevention.
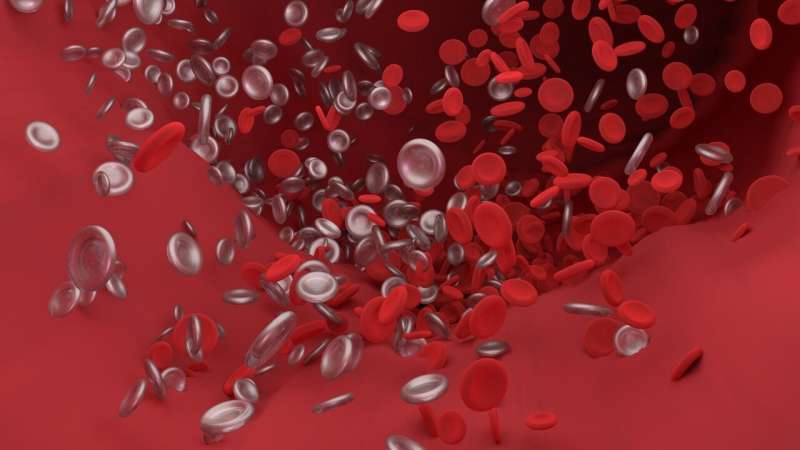
D-dimer is a degradation product of fibrin, which is intimately involved in blood clotting. Fibrin is formed through the action of the protease thrombin on fibrinogen, which causes it to create a polymer. The polymerized fibrin combines with the tiny cells known as platelets and other cellular debris to form a clot.
As a breakdown product, d-dimer is a small protein fragment in the blood after a clot has undergone enzymatic attack through fibrinolysis, a process that attempts to prevent clots from growing. The dimer gets its name, meanwhile, because, chemically, it has two 'D' fragments of the fibrin protein. The amount of d-dimer present in the blood can be determined by a blood test to help diagnose clots.
Thrombosis refers to clot formation, obstructions that have been a pernicious cause of disability and death throughout the pandemic. For example, venous thromboembolism, or VTE, refers to clots that develop in a vein, such as in deep vein thrombosis. Pulmonary embolism refers to a clot that may travel to the lungs, and arterial thromboembolism, or ATE, describes clots that can cause ischemic stroke or myocardial infarction—heart attacks. All have been common among adults hospitalized because of COVID.
"The virus can directly damage the endothelium causing endothelialitis, an immune response within the endothelium in blood vessels, as well as the host response, which in susceptible individuals, is manifested by a hyperinflammatory response and cytokine storm, activating the clotting system as well as platelets," Spyropoulos said. Platelets are the tiny sticky disks that circulate in the blood and help the body form clots. They're a welcome population of cells when plugging a wound—but potentially lethal when coalescing with other factors in the blood to form clots.
The HEP-COVID randomized clinical trial recruited 253 hospitalized adult COVID patients with d-dimer levels more than four times the upper limit of normal. Patients also were recruited into the trial with a diagnosis of excessive clotting induced by sepsis. Patients were studied from May 8, 2020 through May 14, 2021 at 12 academic centers throughout the United States.
Major thromboembolism and death in patients was 28.7 percent for those given a therapeutic-dose of low-molecular-weight heparin, which again, is a high dose measuring four times the current standard of care. Major thromboembolism and death were 41.9 percent for patients receiving standard of care doses, or intermediate-dose heparins, the study found.
"The current standard has been [around] for the past 30 years or so in hospitalized medical patients—including those with pneumonia and sepsis," said Spyropoulos, who worked with an additional international team in an arm of the study reported in the journal Thrombosis Research.
The treatment benefit from low-molecular-weight-heparin therapy was not seen in patients who were critically ill requiring ICU care, a discovery during the research that Spyropoulos said can be understood within the context of the findings. "They were too far advanced in their hyperinflammatory/cytokine storm/coagulopathic state to see any treatment effects by simply increasing heparin dosing."
Regarding the data published in JAMA Internal Medicine, he noted that "HEP-COVID now presents compelling data that we should, in high-risk hospitalized patients with elevated d-dimers, use therapeutic doses of heparins to prevent clots. This is a major change in hospital guidelines."
More information: Alex C. Spyropoulos et al, Efficacy and Safety of Therapeutic-Dose Heparin vs Standard Prophylactic or Intermediate-Dose Heparins for Thromboprophylaxis in High-risk Hospitalized Patients With COVID-19The HEP-COVID Randomized Clinical Trial JAMA Internal Medicine (2021) DOI: 10.1001/jamainternmed.2021.6203
© 2021 Science X Network