Major step forward in development of diagnostic blood test for glioblastoma
Researchers at the University of Sussex are one step further to developing a blood test capable of diagnosing the most aggressive form of brain tumor.
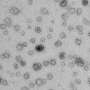
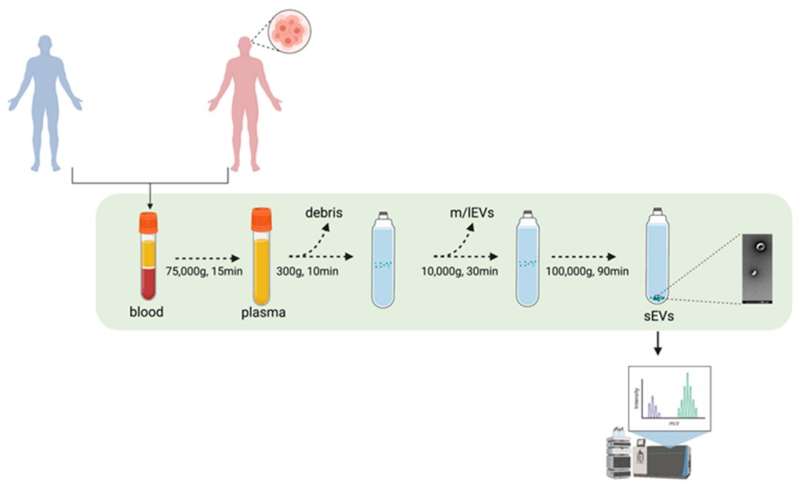
Professor Georgios Giamas and his team, in collaboration with Mr Giles Critchley, Consultant Neurosurgeon and Spinal Surgeon at University Hospitals Sussex, have identified distinctive biomarkers within patient blood samples, which could signal the presence of glioblastoma. The biomarkers (biological signatures for a disease) were identified within extracellular vesicles—small particles which all cells secrete which carry different information, such as DNA or proteins.
The ability to identify these biomarkers within the extracellular vesicles suggests that a liquid biopsy approach could be a viable option for glioblastoma diagnosis, providing both a quicker and less invasive alternative to current diagnostic methods.
More than 11,000 people are diagnosed with a primary brain tumor in the UK each year. Glioblastoma is the most common high grade primary brain tumor in adults, which means it can grow and spread exceptionally quickly. As a result, it's important for diagnosis to be quick, so patients can access treatment as soon as possible.
Georgios Giamas, professor of cancer cell signaling at the University of Sussex, said:
"Currently, glioblastoma detection relies on the display of symptoms, magnetic resonance imaging and invasive tissue biopsies—all of which can delay the identification of the rapidly growing malignant mass.
"A growing body of research is looking into the possibility of developing liquid biopsies which would afford timely and non-invasive assessment of the disease in patients, starting from just a small sample of blood.
"Our study, which effectively does this in a small patient group, is a major step forward in the development of an accurate, non-invasive and time-saving diagnosis method."
In November 2019, Prof Giamas's lab at Sussex identified specific biomarkers that glioblastoma cell lines are able to 'package' within extracellular vesicles allowing for better classification between those cell lines (e.g. less vs more aggressive). Biomarkers can be considered as biological signatures for a disease, and can therefore indicate the presence of cancer in the body. This new stage of research, published in Biomedicines, now proves that a liquid biopsy approach (blood sampling) can successfully determine the presence of such biomarkers in a glioblastoma patient and therefore improve the disease diagnosis and ultimately the patients' prognosis and quality of life.
Prof Giamas now hopes to conduct further research into the clinical value of their findings, by analyzing blood samples of a larger cohort of glioblastoma patients obtained from Genomics England. The team also aims to investigate the presence and levels of such biomarkers in response to specific cancer treatments in an attempt to monitor the progression of the disease and evaluate the importance and role of these biomarkers.
He added: "If we can show that the biomarker signatures of extracellular vesicles obtained from blood samples change/disappear then this could be a huge breakthrough for monitoring the success of treatments too."
More information: Chiara Cilibrasi et al, Definition of an Inflammatory Biomarker Signature in Plasma-Derived Extracellular Vesicles of Glioblastoma Patients, Biomedicines (2022). DOI: 10.3390/biomedicines10010125