Genetic study reveals actionable variation affecting drug efficacy and safety in the Qatari population
A group of researchers at Qatar Foundation have reported the first comprehensive study of the distribution of actionable genetic variation affecting drugs in any Middle Eastern population, with potential implications for preemptive pharmacogenomic implementation in the region. The study has been published online in npj Genomic Medicine.
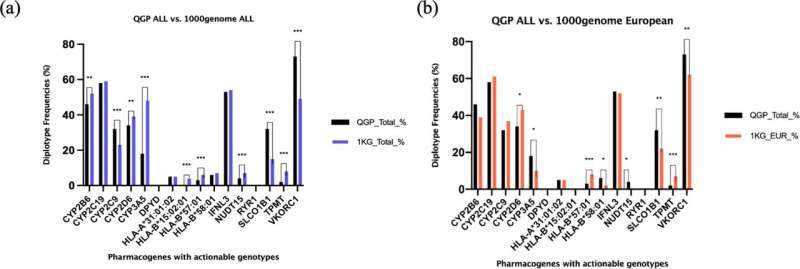
The study, titled "A population study of clinically actionable genetic variation affecting drug response from the Middle East," highlights a diverse distribution of actionable pharmacogenomic variants in the Qatari population compared to other world populations, including a higher frequency in the Qatari population of multiple variants in the gene SLCO1B1, known to increase the risk for muscle toxicity after taking some lipid lowering medications such as simvastatin. By contrast, actionable variation in another gene, CYP3A5, known to affect dosage adjustments to achieve immunosuppression while using the drug tacrolimus was lower in the Qataris compared to other populations.
Pharmacogenomics uses genomic data to predict whether an individual will respond to a specific drug or not. Thus, it helps to move away from a "trial-and-error" way of prescribing medications to being more precise, avoiding medications that are not safe for the patient, but choosing the ones which are more efficacious based on their genetic makeup.
The study led by Dr. Puthen Veettil Jithesh from the College of Health & Life Sciences (CHLS) at Qatar Foundation's (QF's) Hamad Bin Khalifa University (HBKU), along with other scientists from the Qatar Genome Program Research Consortium includes whole genome sequencing data from over 6,000 Qatari individuals.
This data was analyzed using bioinformatics pipelines to identify the frequency distribution of more than 1000 pharmacogenes. Moreover, a detailed and focused analysis was undertaken on 15 genes affecting 46 drugs, which have guidelines for clinical implementation, to understand the clinical impact of actionable genetic variability in the population.
Dr. Jithesh says: "Our findings indicate that 99.5% of the individuals studied have at least one clinically actionable genetic variability with a potential effect on the safety and efficacy of medications. Several of these medications are widely prescribed in Qatar and other parts of the world, and hence our results have important implications in the clinical implementation of pharmacogenomics in the country and beyond. Once validated, patients can be given the right drug in the right dose based on their genetic makeup, without any trial-and-error process. Pharmacogenomics is often considered as the poster child for precision medicine, with its direct clinical applications."
Sir Munir Pirmohamed, David Weatherall Chair of Medicine at the University of Liverpool, and also an author of the paper, says: "This study fills the gap in our understanding of clinically actionable genetic variation affecting medications in the Middle East populations and helps in our planning for precision medicine implementation in the country."
Georges Nemer, professor and interim dean of CHLS says: "This publication is a testimony to our commitment towards robust applied research in the field of precision medicine, and elevates our graduate programs in genomics and precision medicine to higher levels of excellence."
Said Ismail, director of Qatar Genome Program, says: "Based on these research findings we have initiated three pharmacogenomics pilot studies with our partners in the clinics at Hamad Medical Corporation hospital. Such projects are the cornerstone of the implementation of precision medicine in clinical care practice. We, at Qatar Genome Program, are proud to spread head these efforts and will continue this endeavor with larger representation of the population in the near future."
More information: A population study of clinically actionable genetic variation affecting drug response from the Middle East, npj Genomic Medicine, DOI: 10.1038/s41525-022-00281-5