YAP1 molecule essential to development of gastruloids using embryonic stem cells: study
Researchers at the Lewis Katz School of Medicine at Temple University have discovered that the Hippo signaling effector YAP1 is a master regulator of Nodal signaling in human embryonic stem cells (hESCs) for a process essential to human development.
"Our findings may have a wide range of implications, from understanding the process of human development to cancer progression," said Conchi Estarás, Ph.D., Assistant Professor, Cardiovascular Sciences and Center for Translational Medicine at the Katz School of Medicine and a member of the Cancer Epigenetics Institute at Fox Chase Cancer Center.
YAP1 is highly active in development but its activity decreases after birth. However, in adulthood, YAP1 is reactivated in many types of cancer, including liver cancer.
"Our work focuses on understanding the role of YAP in the context of normal stem cells during development. Understanding how YAP1 works in this context will provide us the knowledge necessary to generate new tools to fight against YAP1-driven cancers," said Estarás,
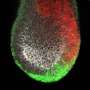
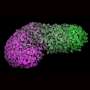
A gastruloid is a small aggregate of cells that mimics an early developmental structure called gastrula. The gastruloid is composed of three layers: ectoderm, mesoderm, and endoderm. These layers contain the founder cells of all organs in the human body, making gastrulation and the gastrula a milestone developmental stage. Errors in this process lead to embryonic death or birth defects.
In order for an embryo to develop past the gastrulation process, the appropriate dose of Nodal protein is essential. Previous research had suggested a role for YAP1 in gastrulation, but more information was needed.
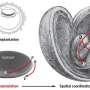
In this study, Estarás and researchers at the Katz School of Medicine and other institutions used hESCs to create gastruloids in vitro and investigate the role of YAP1 in the process of gastrulation. They used 2D micropattern surfaces to make hESCs cells differentiate into mesoderm, ectoderm, and endoderm cells in the so-called gastruloid structure.
"Ethical restrictions forbid the study with human embryos, therefore recreated embryogenesis using in vitro-generated gastruloids allow us to investigate the molecular pathways involved in this essential developmental process," Estarás said.
The analysis yielded two important findings. First, they found that YAP1 regulates the correct allocation of the three-germ layers in the gastruloids and that gastruloids with YAP1 deleted had a reduced ectoderm layer and an enlarged mesoderm and endoderm layers.
"The ectoderm is the germ layer that gives rise to the nervous system and the outer surface of the body, such as the skin," Estarás said. "The right formation of the ectoderm layer is then essential for the development of the embryo."
Second, in order for an embryo to develop past the gastrulation process, the right dose of Nodal protein is essential. Estarás and colleagues found that YAP1 is essential to dictating how much Nodal protein is produced in the gastruloid.
"Removing YAP1 in the gastruloid leads to too much Nodal protein, which in turn leads to gastrulation defects," Estarás said. "In vivo, this mechanism would lead to developmental arrest."
The study, "YAP1 Regulates the Self-Organized Fate Patterning of hESCs-Derived Gastruloids," was published in Stem Cell Reports.
More information: Eleonora Stronati et al, YAP1 regulates the self-organized fate patterning of hESC-derived gastruloids, Stem Cell Reports (2022). DOI: 10.1016/j.stemcr.2021.12.012