Blood vessels regulate obesity through a molecular communication

The abundance and function of blood vessels in adipose tissue conditions the development of obesity. This is the surprising conclusion reached by the study led by laboratories from the Josep Carreras Leukaemia Research Institute and CIC bioGUNE, both members of the Cancer Networking Biomedical Research Center (CIBERONC).
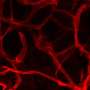
Obesity is a multifactorial epidemic disease closely related to the development of multiple pathologies, originated by the exacerbated expansion of adipose tissue. The development of prevention and treatment strategies for this disease is, therefore, an urgent biomedical need. Adipose tissue has important blood supply requirements, and the contribution of vessels to the development of obesity had not been clarified until now. Dr. Graupera's laboratory is internationally recognized for the study of the regulation and function of blood vessels, addressing the research by using genetically modified animal models.
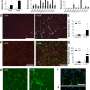
In order to clarify the contribution of blood vessels to the development of obesity, Graupera and Carracedo studied a mouse model with increased vascular functionality, and observed that the animals were resistant to the development of obesity. "At that moment we knew that the vessels were instructing the body to control the management of fat," says Graupera. From this point on, the research team focused its efforts on identifying the mechanism in the vessel's crosstalk with the adipose tissue. "It has been a process in which we have had to turn to cancer research to understand obesity," says Carracedo.
The leaders of this research integrated international experts in different disciplines into the project to provide a comprehensive response to the phenomenon they just described. Years of research led to the conclusion that blood vessels use a language of communication based on small molecules or "metabolites" to instruct adipose tissue. The adipocytes, in response to the message from the vessels, release fat, which the vessels capture as food to proliferate. Dr. Carracedo continues "it is paradoxical that the mechanism by which the vessels produce these metabolites is identical to the one we recently described in the context of prostate cancer." And Graupera adds "while the vessels activate this process during a controlled increase in proliferation, the tumor cells do so in an uncontrolled and aberrant way."
This study sheds light on the importance of blood vessels in regulating health and disease in our body. "Vessels have historically been considered inert blood transport tubes, but we are discovering that they perceive alterations in our body and respond by regulating and organizing the function of multiple tissues." Graupera concludes, "the fact that the vessels respond differently in each organ suggests that we are facing a tissue that is specialized throughout our body, and this opens up exciting opportunities to improve human health through the regulation of blood vessels."
The teams from CIC bioGUNE and the Josep Carreras Leukaemia Research Institute have worked hand in hand with Rubén Nogueiras from CIMUS in Santiago de Compostela, Marc Claret from IDIBAPS in Barcelona and Paul Cohen from the Rockefeller University in New York. The publication and part of the research has received funding from the Fundación BBVA grants program to research teams under the project "Endothelial molecular alterations induced by excessive energy intake—a new concept in obesity and metabolic disorders EndObes."
More information: Erika Monelli et al, Angiocrine polyamine production regulates adiposity, Nature Metabolism (2022). DOI: 10.1038/s42255-022-00544-6