Cryo-EM imaging of STING protein reveals new binding pocket
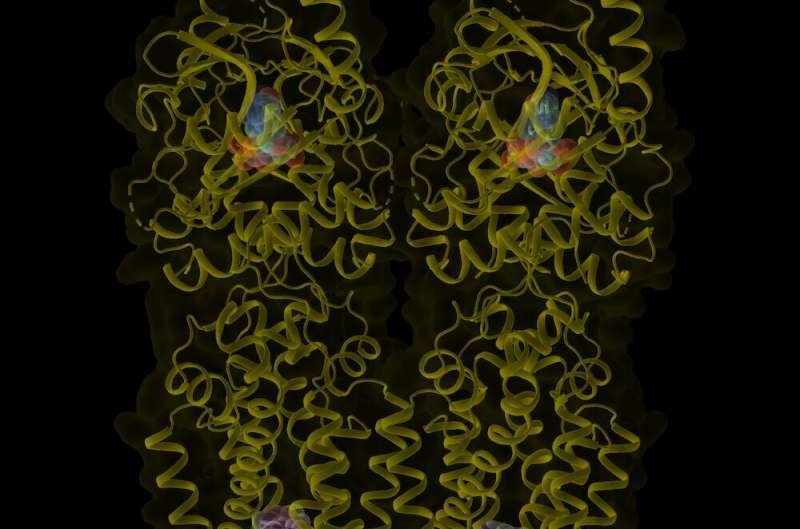
Imaging at near-atomic resolution of a key immune protein commonly known as STING has revealed a previously unrecognized binding site that appears to be pivotal for launching immune attacks, UT Southwestern scientists report in a new study. The findings, published in Nature, could lead to new ways of manipulating STING to prompt stronger immune responses or stem its action in autoimmune diseases.
"For the first time, this work provides a precise picture of the activated state of STING, critical for understanding its role in both normal immunity as well as autoimmune diseases," said study author Xuewu Zhang, Ph.D., Professor of Pharmacology and Biophysics at UT Southwestern. Dr. Zhang co-led the study with Xiaochen Bai, Ph.D., Associate Professor of Biophysics and Cell Biology at UT Southwestern, and their postdoctoral fellows Defen Lu and Guijun Shang. Dr. Zhang and Dr. Bai are members of the Harold C. Simmons Comprehensive Cancer Center.
STING, short for "stimulator of interferon genes," is a central part of the innate immune system, which serves as the body's first line of defense against viruses, bacteria, and cancers. After a sensor known as cGAS detects foreign DNA in cells, it generates a messenger molecule known as cyclic GMP-AMP (cGAMP) that activates STING. In turn, STING launches several signaling pathways that spur the production of inflammatory molecules and chemical signals that prompt cells to clean out detritus to eliminate invaders.
In collaboration with UT Southwestern researcher Zhijian "James" Chen, Ph.D., Professor of Molecular Biology and in the Center for Genetics of Host Defense, the Zhang lab and Bai lab previously reported the first images of STING taken with cryogenic electron microscopy (cryo-EM), a technique that freezes proteins in place to accurately assess their structure, in UTSW's Cryo-Electron Microscopy Facility.
Although this work elucidated some of the fundamental mechanisms that control STING activity, exactly how this protein switches into an active form has been unclear. To answer this question, the Zhang and Bai labs mixed purified STING protein with cGAMP and used cryo-EM to image the resulting product. However, the researchers saw few activated STING molecules, and those that were present were unstable.
Hoping to increase the amount of activated STING available to image, the scientists added an investigational drug known as compound 53 (C53) that's currently being tested as a STING activator for anti-cancer therapy. C53 was assumed to bind to the same site as cGAMP on STING.
The combination of cGAMP and C53 produced significantly more activated STING molecules. But when the researchers searched for C53 on the cryo-EM images, they found it in a completely different location than cGAMP, at the opposite end of the molecule.
"This newly discovered binding site for STING activation came as a complete surprise," Dr. Bai explained. "We call it a 'cryptic pocket' because it appears to form in response to the presence of C53. No evidence of this site exists when C53 is absent."
The fact that STING seems to need both cGAMP and C53 to become strongly and stably activated suggests that an unknown molecule akin to C53 may exist in cells to fill the same role, Dr. Zhang said. Future research will focus on searching for this molecule and better understanding its function.
Someday, the researchers add, drugs that attach to or block this newly discovered binding site could be used to strengthen or dampen immunity to fight infectious or autoimmune diseases.
More information: Defen Lu et al, Activation of STING by targeting a pocket in the transmembrane domain, Nature (2022). DOI: 10.1038/s41586-022-04559-7