Researchers develop long-term in vivo imaging technique to better understand and treat spinal cord injury
A research team led by scientists from the Hong Kong University of Science and Technology (HKUST) has developed an innovative technology for in vivo imaging of the important biological processes involved in the injury and repair of spinal cords, paving the way for a better understanding of the pathology and potential treatment of spinal cord injury (SCI).
A tight bundle of neural cells (neurons and glia) and nerve pathways (axons), the spinal cord serves as a primary information highway between the brain and the peripheral nerves in the rest of our body. Damage to the spinal cord is a devastating and largely irreversible neurological trauma, and can result in lifelong disability and paralysis with no available cure.
While imaging plays an important role in understanding spinal cord functions and its response to pathological insults and therapeutic procedures, there is currently no effective method to capture the injured spinal cord at the level of cellular processes without activating the immune response. Conventional imaging techniques require the patients to have their spinal cord tissue removed to increase image resolution, or run the risk of triggering immune responses in spinal cord tissue, which may affect the disease process being investigated.
Now, a research team led by Prof. Qu Jianan, professor of Department of Electrical & Computer Engineering, and Prof. Liu Kai, associate professor of Division of Life Science at HKUST, has demonstrated a new approach to achieve long-term, repetitive, stable, high-resolution, and inflammation-free in vivo spinal cord imaging in mouse models.
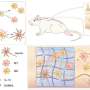
In their proposed protocol, ligamentum flavum (LF)—the ligaments connecting adjacent vertebrae in our spine—is retained to protect the underlying spinal cord tissue and reduce the risk of imaging window activating inflammation. But retaining the LF layer also means sacrificing the imaging quality, because the layer introduces optical scattering and results in decreased penetration depth of spinal cord imaging.
To solve this problem, the team applied iodixanol, an FDA-approved non-toxic compound, as an optical clearing medium for the imaging window and greatly enhanced its transparency as well as image contrast and resolution. Compared with the prior methods, the iodixanol-based optical clearing technique allows the researchers to remove less tissue above the spinal cord without compromising imaging quality, thus significantly extending the number of imaging sessions to up to 15 sessions over 167 days.
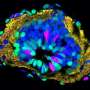
Using this optically cleared intervertebral window, the team studied neuron-glia dynamics and observed strengthened contact of microglia with the nodes of Ranvier during axonal degeneration, opening a promising way to study the interaction between immune cells and nodes of Ranvier under normal and injury conditions. The results were recently published in Nature Communications.
"Considering the difficulties associated with long-term and repetitive spinal cord imaging, this innovation will be an important and widely used tool for the study of spinal cord injury," said Prof. Qu, who is an expert of optical engineering and science with extensive experience in in vivo linear and nonlinear optical spectroscopy and imaging of biological tissues from a variety of animal models.
"By avoiding surgery-induced inflammation, we can track microglia from resting to activation stages and understand its functional interaction with degenerating and regenerating axons in the spinal cord," added Prof. Liu, whose research interests include the cellular and molecular mechanisms of axonal regeneration in the adult mammalian central nervous system. "In vivo imaging in living animal models will reveal new biological insights leading to efficient therapeutic strategies for SCI treatment."
More information: Wanjie Wu et al, Long-term in vivo imaging of mouse spinal cord through an optically cleared intervertebral window, Nature Communications (2022). DOI: 10.1038/s41467-022-29496-x