Slow pace for youngest kids getting COVID vaccine doses
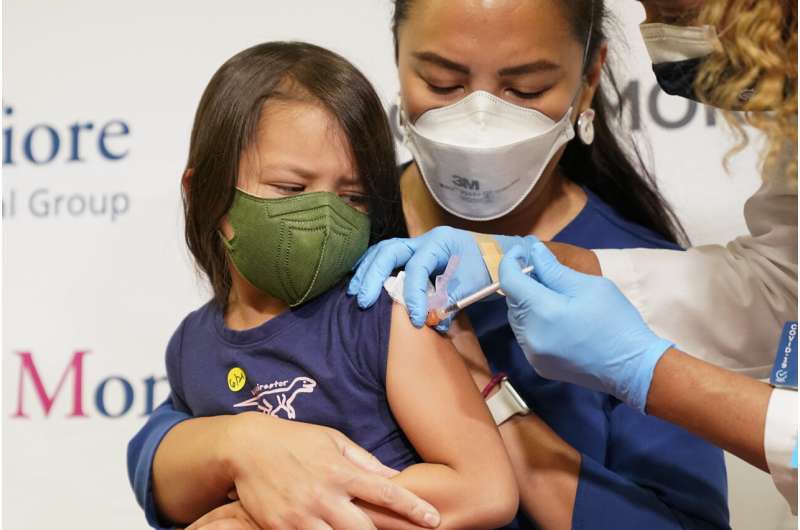
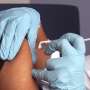
Nearly 300,000 children under 5 have received COVID-19 shots in the two weeks since they became available, a slower pace than for older groups. But the White House says that was expected for the eligible U.S. population of about 18 million kids.
The Centers for Disease Control and Prevention was to publish initial data on shots for the age group later Thursday, reflecting doses administered since regulators authorized them on June 18. The first vaccinations didn't begin until several days later because the doses had to be shipped to doctors' offices and pharmacies.
U.S. officials had long predicted that the pace of vaccinating the youngest kids would be slower than for older groups. They expect most shots to take place at pediatricians' offices.
Many parents may be more comfortable getting the vaccine for their kids at their regular doctors, White House COVID-19 coordinator Dr. Ashish Jha told The Associated Press last month. He predicted the pace of vaccination would be far slower than it was for older populations.
"We're going see vaccinations ramp up over weeks and even potentially over a couple of months," Jha said.
Officials also note there is some hesitance about the need for shots for kids who are far less likely than older age groups to develop serious illness or die from the coronavirus. Parents are being encouraged to talk to the child's doctor for trusted information about the benefits of the vaccines.
A Kaiser Family Foundation survey conducted in April found that 1 in 5 parents of children under age 5 said they would get their child vaccinated right away once it was authorized, 38% said they would wait and see, and nearly 4 in 10 said they wouldn't get their children vaccinated at all or only if required.
More than 5 million pediatric doses have been shipped to more than 15,000 locations, the White House said, ready for parents and kids to come in.
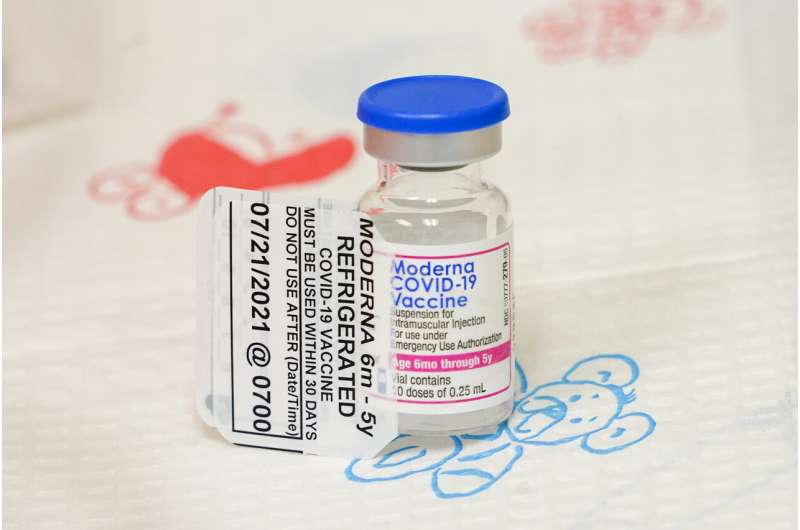
Still, the roll-out hasn't been without hiccups for some parents of babies and toddlers, because of state restrictions on pharmacies and because some doctors are not offering the shots.
Many states bar pharmacies from vaccinating children under 3 years old or require prescriptions for them to do so. In other cases, pharmacies have inadequate staff, space or training to give the shots correctly to the youngest, said Allie Jo Shipman, director of state policy for the National Alliance of State Pharmacy Associations.
"It's just a more complex situation than it is with your older children and adults," Shipman said.
Some doctors opted not to offer the shots because of cold storage requirements or concern about wasting doses. The vaccine comes in multi-dose vials that must be used within 12 hours after opening.
"They don't want to see vaccine go unused," said West Virginia pediatrician Dr. Lisa Costello, who is working with her state's COVID-19 response task force. "You either need to find the number of people to vaccinate for that vial or discard the doses that are left over."
Organizing vaccine times after hours or clustering vaccine appointments are possible solutions doctors are trying, Costello said. And West Virginia has encouraged doctors to go ahead and open the vials even if they have only one patient to vaccinate, she said.
For older children, those 5 to 11 years old, vaccine coverage has varied widely from state to state, from a low of 11% of that age group fully vaccinated in Alabama to 63% in Vermont, according to an analysis of vaccination data through June 29 by the Kaiser Family Foundation.
Of the top 10 states for vaccination coverage in the 5 to 11 age group, five are in New England. Of the bottom 10 states, nine are in the South.
The Biden administration said that while the slow pace of vaccination in the youngest group was expected, officials won't be satisfied until as many people as possible receive the protection of vaccines. Parents can use vaccines.gov to search for vaccine providers by location, vaccine brand and age groups vaccinated.
© 2022 The Associated Press. All rights reserved. This material may not be published, broadcast, rewritten or redistributed without permission.