This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Covid treatment gaps highlight persistent health care challenges
Millions of Americans don't have easy access to facilities that dispense oral antiviral treatments for COVID-19, according to a new Yale study. In some parts of the country, researchers found, people have to travel 30 minutes or more to get them.
The results, they say, highlight persistent challenges to health care access in parts of the United States and larger disparities in care in the U.S. health care system.
The findings were published Jan. 3 in JAMA Network Open.
Two oral medications have received emergency use authorization from the U.S. Food and Drug Administration (FDA) for treatment of COVID-19—nirmatrelvir-ritonavir, branded as Paxlovid, and molnupiravir, branded as Lagevrio.
"For many patients diagnosed with COVID-19, physicians recommend oral therapies," said Peter Kahn, a pulmonary and critical care fellow in Yale School of Medicine and lead author of the study. The drugs can prevent the development of severe COVID-19 in specific populations and there's some evidence the therapies may prevent long COVID.
Until recently, monoclonal antibody infusions were recommended for some cases, but on Nov. 30, the FDA rescinded its authorization of the last monoclonal antibody treatment as these therapies proved ineffective at treating newer variants of SARS-CoV-2, the virus that causes COVID-19.
"With monoclonal antibodies no longer an option for treatment of COVID-19, oral therapies and remdesivir, which is administered intravenously, are the remaining choices for patients," said Kahn. "Understanding how accessible they are to patients becomes even more important."
For the study, the researchers used publicly available data to assess travel times to facilities in the United States that dispense COVID-19 oral therapies. They used driving time rather than distance to quantify accessibility in order to better capture real-world scenarios.
"When we think about traveling somewhere, to the nearest drug store, for example, we tend to think about how long it will take us to get there, not the distance between the two points," said Kahn. "For this work, we wanted to add additional nuance and insight into accessibility by thinking about the way in which patients experience access."
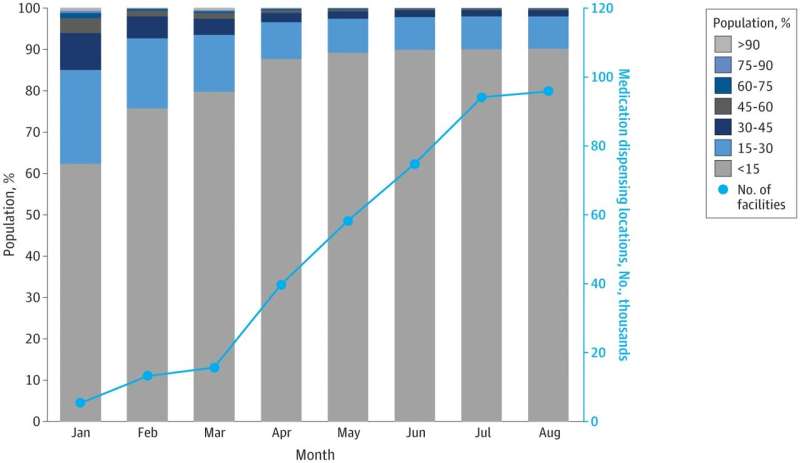
Paxlovid and Lagevrio were granted emergency use authorization in December 2021 and rollout began quickly. Kahn and his colleagues found that in January 2022, 62% of the U.S. population were within 15 minutes of the nearest oral therapy dispenser. By May 2022, around 90% percent of U.S. residents were within 15 minutes of dispensers. That percentage has remained steady ever since, despite thousands of additional locations being added since May, leaving more than 6 million people more than 30 minutes away from distributers of oral COVID-19 therapies.
"Distribution of these oral therapies was efficient," said Kahn. "The federal government started in key areas that had the widest spectrum of access and then ramped up distribution to secondary sites that provided greater coverage as rapidly as possible. The areas that still lack access, in many ways, speak to the structural barriers to health care access that have existed for some time."
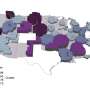
When the researchers mapped drive times to COVID-19 oral therapies, they found that the United States was almost divided in half. In the eastern half of the country, oral therapies were readily available with scant few areas averaging longer drive times. But in the western half, there were many more areas where patients had to drive 30 minutes or longer to get medication. Those living farthest from their nearest dispensing location—an hour or more away—were largely concentrated in South Dakota, North Dakota, Montana, Texas and Nevada.
"There are pockets in the western part of the country where access to oral therapies remains a challenge," said Kahn. "Our findings highlight areas where the population is underserved and links the cracks exposed in the pandemic to larger questions on procuring health care access for the millions of individuals in these areas. These disparities will likely persist without thoughtful health care delivery innovation."
These access challenges pose a problem both for health care in general and COVID-19 mitigation in particular, he added.
"Ultimately, one of the many lessons COVID-19 taught is how interconnected we all are, both as a country and as a global society," Kahn said. "Every individual is impacted by health care access challenges, whether in our backyard, across the country, or across the globe."
Kahn and his colleagues hope the findings encourage policymakers and health care groups to consider how to address these persistent gaps in access, both in general and specifically for COVID-19 treatment, where early use is most effective.
"The question we have to ask is to what extent we're OK with these areas continuing to experience access challenges," said Kahn, "and how as a national and global community we can innovate to come up with novel ways to deliver better and attainable health care to our neighbors across the country and across the world."
More information: Peter A. Kahn et al, Travel Times to Facilities Offering Oral Medication Therapy for COVID-19, JAMA Network Open (2023). DOI: 10.1001/jamanetworkopen.2022.49810