This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
New dual-phase imaging protocol reduces costs and radiation exposure for cognitively impaired patients
A novel nuclear medicine imaging protocol can take the place of two separate imaging scans for the evaluation of brain changes linked to cognitive impairment, cutting costs and radiation exposure for patients as well as providing them greater comfort.
The protocol—known as dual-phase amyloid PET—can assess both amyloid deposition and neurodegeneration with a single tracer injection, assisting physicians as they classify patients on the Alzheimer's disease continuum. This research was published in the February issue of The Journal of Nuclear Medicine.
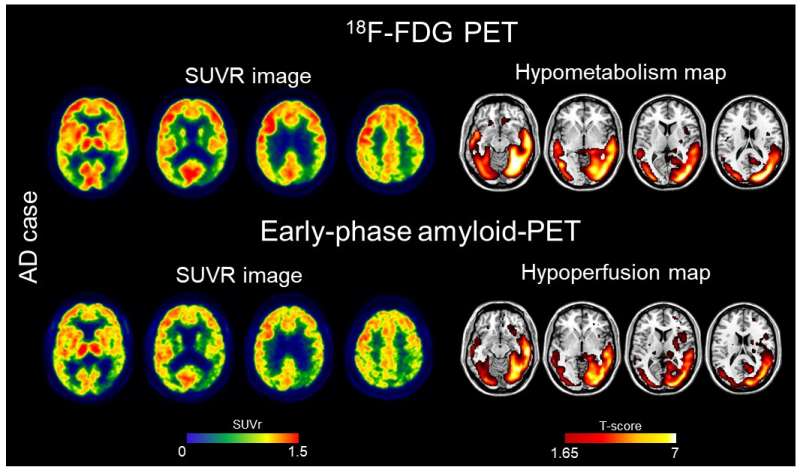
Alzheimer's disease causes several changes in the brain, including neurodegeneration and beta-amyloid deposition. Currently, 18F-FDG scans are used to measure brain metabolism, which can indicate neurodegeneration. Amyloid PET scans are performed to evaluate beta-amyloid deposition, a key characteristic of Alzheimer's disease.
"During the amyloid PET scans, nuclear medicine physicians wait at least 50 minutes after the radiotracer injection to acquire the image of beta-amyloid deposition. If images are also taken immediately after injection (early-phase), they can provide a measure of brain perfusion. Since brain perfusion and brain metabolism are closely related, the perfusion information could replace an 18F-FDG PET scan," said Cecilia Boccalini, MSc, Ph.D. candidate at the Laboratory of Neuroimaging and Innovative Molecular Tracers and faculty of medicine at the University of Geneva Neurocenter in Geneva, Switzerland and faculty of neuroscience at Vita-Salute San Raffaele University in Milan, Italy.
The study aimed to compare early-phase amyloid PET scans and 18F-FDG scans at the individual level, as well as their ability to distinguish patients along the Alzheimer's disease continuum. A total of 166 patients ranging from cognitively unimpaired to mild cognitive impairment and dementia underwent 18F-FDG and early-phase amyloid PET scans. Brain hypoperfusion maps (from the early-phase amyloid PET) and brain hypometabolism maps (from 18F-FDG PET) were created and compared for each patient.
Results showed that brain hypoperfusion assessed by early-phase amyloid PET is comparable with brain hypometabolism assessed by 18F-FDG-PET. Hypoperfusion and hypometabolism patterns were also equally able to distinguish patients with neurodegenerative diseases from controls.
"This dual-phase amyloid-PET protocol allows physicians to measure brain amyloid and perfusion in a single procedure," stated Valentina Garibotto, MD, head of the Nuclear Medicine and Molecular Imaging Division at Geneva University Hospitals in Geneva, Switzerland, and associate professor at the University of Geneva.
"Obtaining both amyloid and perfusion data with a single tracer injection is optimal in terms of patient safety and radiation exposure, comfort, and costs. Our work supports the routine use of the dual-phase amyloid PET protocol in clinical practice."
More information: Cecilia Boccalini et al, Early-Phase18F-Florbetapir and18F-Flutemetamol Images as Proxies of Brain Metabolism in a Memory Clinic Setting, Journal of Nuclear Medicine (2022). DOI: 10.2967/jnumed.122.264256