This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
reputable news agency
proofread
FDA authorizes combination flu-COVID test for home use

The Food and Drug Administration on Friday approved the first combination test for flu and C OVID-19 that can be used at home, giving consumers an easy way to determine if a runny nose is caused by either disease.
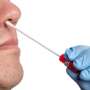
The Lucira COVID-19 & Flu Home test, which can be purchased without a prescription, uses self-collected nasal swab samples and delivers results in about 30 minutes, the agency said.
While at-home COVID tests are readily available, this is the first home test for influenza A and B, commonly known as the flu. The test was granted an emergency use authorization, which facilitates the availability of "medical countermeasures" during public health emergencies.
Jeff Shuren, director of the FDA's Center for Devices and Radiological Health, called the authorization "a major milestone in bringing greater consumer access to diagnostic tests that can be performed entirely at home."
The agency said the test is for individuals "with signs and symptoms consistent with a respiratory tract infection" and said it can be used on children as young as 2, with adults collecting the samples.
It recommends that tests be reported to healthcare providers and cautions that there is a risk of false positive and negative results. "Individuals who test negative and continue to experience symptoms of fever, cough and-or shortness of breath may still have a respiratory infection and should seek follow-up care with their healthcare provider," the agency said.
Citing the impact of COVID and RSV, another respiratory infection, the FDA said it "recognizes the benefits that home testing can provide" and would work to increase the number of tests available.
© 2023 The Associated Press. All rights reserved. This material may not be published, broadcast, rewritten or redistributed without permission.