FDA authorizes first OTC COVID-19 Home Test Collection Kit
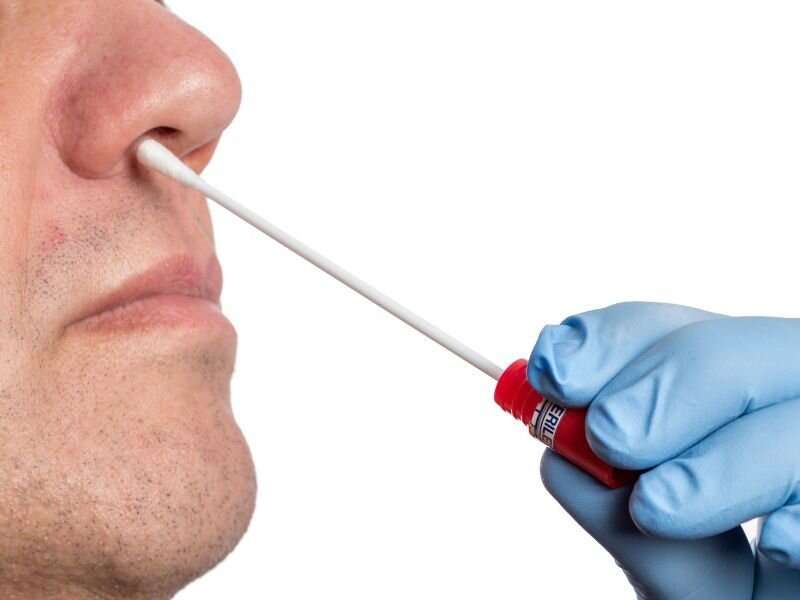
(HealthDay)—The U.S. Food and Drug Administration authorized the first over-the-counter home sample collection kit for COVID-19, the agency announced yesterday.
The LabCorp Pixel COVID-19 Test Home Collection Kit, authorized for individuals 18 years and older, allows for self-collection of a nasal swab at home that can then be sent for testing at LabCorp. Users then receive their test results via a phone call from their health care provider or by email or online portal, depending on whether the results are positive, negative, or invalid.
The use of a home sample collection kit allows users to "access information about their COVID-19 infection status that could aid with determining if self-isolation (quarantine) is appropriate and to assist with health care decisions after discussion with a health care professional," the FDA notes.
"While many home collection kits can be prescribed with a simple online questionnaire, this newly authorized direct-to-consumer collection kit removes that step from the process, allowing anyone to collect their sample and send it to the lab for processing," Jeff Shuren, M.D., J.D., director of the FDA Center for Devices and Radiological Health, said in an agency news release.
On Dec. 4, the FDA also announced the authorization of a diagnostic test for at-home collection of samples to detect both COVID-19 and influenza A and B. The Quest Diagnostics RC COVID-19 +Flu RT-PCR Test is available by prescription with the Quest Diagnostics Self-Collection Kit for COVID-19 +Flu for patients with suspected respiratory viral infection with COVID-19. Patients can ship their samples to a Quest Diagnostics laboratory for analysis.
More information:
More Information - LabCorp Test
More Information - Quest Diagnostics Test
Copyright © 2020 HealthDay. All rights reserved.